Three months ago, I wrote about how the Cleveland Clinic had recently opened a clinic that dispensed herbal medicine according to traditional Chinese medicine (TCM) practice. As regular readers might expect, I was not particularly impressed or approving of this particular bit of infiltration of quackademic medicine into a major, generally well-respected academic medical center, particularly given some of the amazingly pseudoscientific treatments espoused by the naturopath who was running the clinic. I also pointed out that, although herbalism is the most plausible (or perhaps I should say the least implausible) of modalities commonly associated with “complementary and alternative medicine” (CAM) or “integrative medicine”, it still exhibits a number of problems, the biggest of which is what I like to call either the delivery problem or the bioavailability problem. In brief, herbs, when they work, are adulterated drugs. The active ingredient is usually a minor constituent, embedded in thousands of other constituents that make up herbs, and it’s almost impossible to control lot-to-lot consistency with respect to content or active ingredients given how location, weather, soil conditions, rainfall, and many other factors can affect how the plants from which the medicines are extracted grow and therefore their chemical composition. To demonstrate the concept, I pointed out that it’s much safer and more predictable to administer digoxin to a patient who needs its activity on the heart than it would be for the patient to chew on some foxglove leaves, given that the therapeutic window (the difference between the doses needed to produce therapeutic effects and the lowest dose that will cause significant toxicity) is narrow.
Which brings me to medical marijuana, a.k.a. medical cannabis.
Before I continue, let me just state my position on marijuana, which is different than it was, say, 20 years ago. Today, I believe there’s no reason why marijuana shouldn’t be legalized and treated by states the same way as tobacco products and alcoholic beverages are; they should be heavily regulated and taxed. Among physicians, this appears to be a common view, at least if you can believe a poll I saw a while back (for which I can’t find the link, alas). It’s also, these days, more and more of a mainstream view. In any case, medical marijuana has been a topic I’ve been meaning to write about for a while, now, but my “Dug the Dog” tendencies have kept popping up over squirrels topics like the Food Babe, ketogenic diets for cancer, and a variety of other topics.
Medical marijuana arrived in my state in 2008, when the voters approved a measure permitting it. After some time for the state to draft regulations, the law was implemented, and I had the strange (to me at the time) experience of receiving notices about state regulations, requirements, and documentation should I wish to prescribe medical marijuana. Indeed, more than twenty states, plus the District of Columbia, have legalized medical marijuana. They’ve done so on the basis of a political movement among patients that make pot sound like a miracle drug that can help when no other intervention can. And it’s more than that. Medical cannabis has been touted as a near-panacea for everything from pain to chemotherapy-induced nausea to HIV- and cancer-induced cachexia to even curing cancer itself. Yes, there’s a lot of hype out there, and there are a lot of claims that sometimes go viral on various social media, even though the evidence to support the claims is often, to put it mildly, less than rigorous.
Indeed, the acceptance of medical marijuana appears to be far more driven by politics than it is by science, as was pointed out in a recent New York Times article about the impending legalization of medical cannabis in New York State:
New York moved last week to join 22 states in legalizing medical marijuana for patients with a diverse array of debilitating ailments, encompassing epilepsy and cancer, Crohn’s disease and Parkinson’s. Yet there is no rigorous scientific evidence that marijuana effectively treats the symptoms of many of the illnesses for which states have authorized its use.
Instead, experts say, lawmakers and the authors of public referendums have acted largely on the basis of animal studies and heart-wrenching anecdotes. The results have sometimes confounded doctors and researchers.
I note that this article was written over a week before the Governor signed New York’s medical marijuana bill into law, thus legalizing it in New York. The article then goes on to give several examples, such as Alzheimer’s disease, lupus, Sjogren’s syndrome, Tourette’s syndrome, Arnold-Chiari malformation and nail-patella syndrome, and in particular rheumatoid arthritis:
Yet there are no published trials of smoked marijuana in rheumatoid arthritis patients, said Dr. Mary-Ann Fitzcharles, a rheumatologist at McGill University who reviewed the evidence of the drug’s efficacy in treating rheumatic diseases. “When we look at herbal cannabis, we have zero evidence for efficacy,” she said. “Unfortunately this is being driven by regulatory authorities, not by sound clinical judgment.”
As is the case with so much herbalism—and, make no mistake, medical marijuana is the new, popular herbalism of the moment—claims have far outstripped the evidence. Also, as pointed out in the NYT article, even advocates of medical marijuana admit that “the state laws legalizing it did not result from careful reviews of the medical literature.”
That’s the understatement of the year! Reading advocate websites for medical marijuana, I ask myself if there’s any disease or condition the holy weed isn’t good for? Truly, we are talking belief over science!
Unfortunately, even famous doctors like Sanjay Gupta are getting in on the act with a report, “Cannabis Madness,” full of a lot of anecdotes and rhetoric about “policy against patients.” Again, I believe that marijuana should be legalized, regulated, and taxed, just like alcohol and tobacco. If marijuana is going to be approved for use as medicine rather than for recreational use, however, the standards of evidence it must meet should be no different than any other drug, and for the vast majority of indications for which it’s touted medical cannabis doesn’t even come close to meeting that standard.
The evidence
There are definitely chemicals with potential medicinal use in the marijuana. No one, even the most die-hard drug warrior, denies that. These compounds are called cannabinoids, which is a term that describes a family of complex molecules that bind to cannabinoid receptors, which are proteins on the surface of cells. There are two types of cannabinoid receptors, type 1 (CB1) and type 2 (CB2). These receptors are seven transmembrane G-protein coupled receptors (so named for the seven protein domains that span the membrane), a class of receptor I’m pretty familiar with, because one of the receptors I study is of the same class, which looks like this:
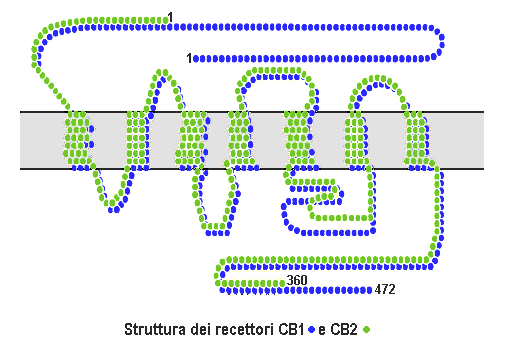
“Cb1 cb2 structure” by Esculapio at it.wikipedia – Own work (Original caption: “Immagine creata da —Esculapio”). Licensed under CC BY-SA 3.0 via Wikimedia Commons.
The details of how this happens aren’t essential for this particular post, but when these receptors are stimulated by the binding of cannabinoid molecules, including endocannabinoids (produced by mammals), plant cannabinoids (such as (−)-trans-Δ9-tetrahydrocannabinol, more commonly referred to by its abbreviation THC) produced by (for example) the cannabis plant, and synthetic cannabinoids (such as HU-210), downstream chemical signaling pathways are initiated from the receptor to the inside of the cell, thus producing the effects on the cell and organism. There is mounting evidence that there are more than two types of cannabinoid receptors. In any case, CB1 receptors are found widely in the central nervous system, where they modulate a variety of responses, and are also found in other parts of the body, for instance, the pituitary gland, thyroid gland, lungs, and kidney, as well as fat cells, muscle cells, liver cells, and in the digestive tract. CB2 receptors, on the other hand, are expressed primarily in the immune system, the gastrointestinal tract, and, to a much lesser extent than CB1 receptors, in the brain and have been implicated in modulation of immune responses. In particular, stimulating CB2 receptors cannabinoids could be potentially useful as anti-inflammatory drugs. Over the last couple of decades, endocannabinoids and cannabinoid receptors have been implicated a large variety of functions, including memory, pain, energy metabolism, and more. It is thus plausible that manipulation of cannabinoid signaling could have therapeutic effects in a variety of areas.
Unfortunately, one of the problems with medical marijuana, as noted in the NYT article is that enthusiasm for weed as a cure-all has far outstripped existing medical evidence. This disconnect between the existing evidence base ranges from thin to nonexistent, depending on the condition. One of the most frequent claims I see is that cannabis can be used to treat cancer. I’m not going to address that claim specifically in this post, except very briefly, because I think it’s a large enough topic to warrant its own post. Suffice to say that interesting preclinical studies have been exaggerated beyond all evidence, but nonetheless certain cannabinoids could have potential in the treatment of certain cancers. I might also review the evidence base for cannabinoids and autism, given how I’ve been seeing discussions of its use starting to pop up lately on the usual sites. In other words, stay tuned for parts two and three spread out over the next several weeks, whenever no squirrels distract Dug the Dog.
In all fairness, in this country, at least, studying the medicinal properties of marijuana and its constituents is not easy, given that it is currently an illegal drug, as was discussed in the NYT article. It’s not for lack of interest, but mainly because the law (and therefore the Drug Enforcement Agency) classifies it as a schedule 1 drug with “no currently accepted medical use.” Scientists who want to do research on marijuana and its constituents—particularly clinical trials—must register with the DEA and submit an investigational new drug (IND) application to the Food and Drug Administration for human trials. Moreover, the National Institute on Drug Abuse is the only supplier of legal, research-grade marijuana. On the other hand, while doing research on marijuana is difficult in this country, researchers in other countries that have long had much more lax laws and regulations should have an easier time of it.
Another issue is how to do a proper placebo control. Given that many of the conditions for which medical marijuana is touted are conditions with a large subjective symptomatic component, such as pain, nausea, fatigue, or lack of appetite, clinical studies of medical marijuana are going to require really good placebo controls. Given that at least one of the active components causes a high, it’s arguably even more difficult than in the case of, for instance, acupuncture, to design studies with adequate controls. That’s why most of the more rigorous studies have used specific purified cannabinoids. For example, in this study, a titanium pipe loaded with doses of THC varying potencies is used rather than plant, while this study of cannabis for neuropathic pain used high-dose cannabis, low-dose cannabis, and placebo cigarettes.
Be that as it may, let’s look at the evidence base for conditions for which medical marijuana might provide a benefit. Remember, again: I’m leaving out cancer and autism for another day. Leaving these aside, here are the potential medical uses for marijuana for which evidence exists that ranges from reasonably good to suggestive.
Chronic pain. It’s been known for a long time that cannabinoids modulate pain responses; so it’s plausible that either smoked marijuana or cannabinoids isolated from marijuana (or synthetic cannabinoids) could be useful for chronic pain. Fortunately, this is one of the more widely-studied uses for medical cannabis. For example, a recent review of uses of cannabinoids for the treatment of non-cancer pain concluded that there was evidence that cannabinoids are safe and modestly effective in neuropathic pain, citing preliminary evidence of efficacy in fibromyalgia and rheumatoid arthritis. As is the case with most reviews, more study was recommended. This particular review included smoked cannabis, oromucosal extracts of cannabis based medicine, nabilone (a synthetic cannabinoid), dronabinol (a synthetic delta-9-THC), and a novel THC analogue. Most studies have only been short term, and adverse events have tended not to be serious. The current general recommendation is that cannaboids should probably not be used as first line agents “for conditions for which there are more supported and better-tolerated agents,” and adverse effects are not well studied.
Appetite stimulation. I’ve never smoked marijuana, but those who have, have told me about the “munchies,” something that anyone who’s ever seen a comedy in which characters smoke post has likely seen used as fodder for jokes. Given its ability to stimulate appetite, it is therefore plausible that medical cannabis might be useful for appetite stimulation in patients with cachexia due to cancer or HIV/AIDS. (Cachexia is the “wasting” that can occur in advanced cases of malignancy and AIDS, among other diseases.) Unfortunately, a recent Cochrane review noted variable outcomes and concluded that the “efficacy and safety of cannabis and cannabinoids in this setting is lacking” and noting no good evidence of long-term effects on AIDS-related mortality and morbidity. Regarding cancer cachexia, Peter Lipson noted several years ago a study that failed to find any benefit from cannabis extract for cancer-related cachexia, speculating that maybe the mechanisms that cause appetite suppression in cancer are different than the mechanisms by which cannabinoids modulate appetite.
Currently, there are few controlled trials cited at the NCI website, which, taken together, find that oral THC has variable effects on appetite stimulation and weight loss in patients with advanced malignancies and human immunodeficiency virus (HIV) infection. A PubMed review by yours truly also found the evidence rather sparse. For instance, this randomized trial testing cannabis extract (CE), THC, and placebo (PL) reported that “no differences in patients’ appetite or quality of life were found either between CE, THC, and PL or between CE and THC at the dosages investigated.” Another randomized trial comparing megestrol acetate (Megase) and dronabinol found that “megestrol acetate provided superior anorexia palliation among advanced cancer patients compared with dronabinol alone” and that “combination therapy did not appear to confer additional benefit.” A more recent small randomized trial tested THC versus placebo and found that “THC may be useful in the palliation of chemosensory alterations and to improve food enjoyment for cancer patients.” To be honest, I was shocked at how sparse the literature is covering this particular indication. Indeed, as the NCI notes, there are no randomized controlled trials of smoked cannabis for this indication in cancer patients.
Nausea/antiemetic. Despite many advances in anti-emetics (anti-nausea and vomiting) agents, cancer-induced nausea and vomiting (CINV) is still among the most troubling symptoms cancer patients face. There are two FDA-approved cannabis products for this indication, dronabinol and the synthetic cannabinoid nabilone. The NCI cites several clinical trials and meta-analyses finding that these two drugs are efficacious against CINV, and the National Comprehensive Cancer Network guidelines recommend these drugs as treatment for breakthrough nausea and vomiting due to chemotherapy. One systematic review from 2001 found that cannabinoids were slightly more effective antiemetics than prochlorperazine, metoclopramide, chlorpromazine, thiethylperazine, haloperidol, domperidone, or alizapride, but were not more effective in patients already using large doses of antiemetic drugs. A more recent systematic review and meta-analysis found that cannabinoids were superior to conventional drugs but that “adverse effects were more intense and occurred more often among patients who used cannabinoids.” In children with cancer undergoing chemotherapy, a Cochrane systematic review concluded that “cannabinoids are probably effective but produce frequent side effects” and that the review “suggests that 5-HT(3) [seratonin] antagonists with dexamethasone added are effective in patients who are to receive highly emetogenic chemotherapy although the risk-benefit profile of additional steroid remains uncertain.”
Inflammatory bowel disease (IBD). Last fall, the first clinical trial of cannabis in IBD was reported by a group of Israeli researchers. It was a small trial (21 patients), in which subjects were assigned randomly to groups given cannabis, twice daily, in the form of cigarettes containing 115 mg of Δ9-tetrahydrocannabinol (THC) or placebo containing cannabis flowers from which the THC had been extracted. A clinical response was achieved in 10 of 11 patients receiving cannabis with THC and 4 of 10 in the placebo group. Overall, this was a small study, but intriguing. No difference in complete remissions between the groups was observed, but that could easily be because of the small numbers. As with many conditions, all one can conclude is that more research is needed.
There is, of course, a laundry list of other conditions. Cannabinoids have been shown to lower intraocular pressure, making them potentially useful for treating glaucoma, although using cannabis to treat glaucoma is impractical in the vast majority of patients (see below), and there exist better treatments. After that, other conditions for which medical cannabis is frequently recommended include schizophrenia, for which a Cochrane Review concludes that there is no good evidence for or against the use of cannabis for schizophrenia. For epilepsy, data from double-blind randomized controlled clinical trials is lacking, although clinical trials are finally being done.
Overall, the evidence base supporting medical cannabis use, from my interpretation, ranges from nonexistent (most indications) to suggestive (e.g., anti-inflammatory), to fairly good in one case (ant-emetic). However, most of the good clinical trials didn’t use marijuana cigarettes as most patients get them, but rather either purified cannabinoids (or synthetic analogues) or cannabis cigarettes spiked with varying amounts of THC. Indeed, all of these studies tend to suggest that purified drugs from cannabis or synthetic drugs based on compounds designed to mimic either endocannabinoids or cannabinoids from marijuana will be the future. I realize that that’s not what medical marijuana activists want to hear. I also realize that it is likely I will be lambasted as a “pharma shill” or as so “conventional” that I can’t think outside the box, but I’ve endured those attacks before when I’ve criticized other forms of herbalism—and, make no mistake, medical marijuana is herbalism. In any case, mine, I believe, is a reasonable interpretation of the currently existing medical literature.
Moreover, contrary to what advocates will claim, cannabis, particularly smoked cannabis, is not without adverse health effects, as was recently reviewed in the New England Journal of Medicine. Potential medical effects reported in long time users include motor vehicle collisions (not unreasonable to expect because driving while high is not a good idea), chronic bronchitis (not surprising as a result of smoke inhalation), schizophrenia (one wonders whether correlation really suggests causation here), depression, and addiction to other drugs, although the risk for cancer due to marijuana smoke appears to be much lower than with tobacco cigarettes. True, drug warriors and moralists will frequently exaggerate the risks in order to promote their agendas, but that doesn’t mean that cannabis is perfectly safe and doesn’t produce significant side effects or complications.
Then there’s the delivery problem.
Delivery, purity, highs
Let’s consider, for a moment, a generic herb that has medicinal properties. I began this post by briefly discussing the problems with herbs as medicine, but I didn’t discuss delivery. If one were to come up with a delivery method for an effective herb, one would be hard pressed to come up with a worse method than burning it and inhaling it. Consider the case of tobacco. The combustion of dried tobacco leaves produces a toxic stew of gases with carcinogenic effects. Of course, the main reason tobacco is so addictive is because it does have an active drug in it, specifically nicotine, which rapidly reaches the circulation through the alveolar sacs in the lungs. However, that nicotine is mixed with numerous combustion products that can cause cancer and contribute to the numerous other diseases to which smoking tobacco has been linked.
This brings us back to delivery. People have been using marijuana for the high and for medicinal purposes for a very long time, but cannabinoids were only first isolated from the plant in the 1940s, and the main active ingredient, (−)-trans-Δ9-tetrahydrocannabinol (THC), wasn’t discovered until the 1960s. Now, like the case with cigarette smoke and its delivery of nicotine to the bloodstream, the THC and other active cannabinoids delivered to the bloodstream through smoking marijuana are mixed in a similarly toxic stew of combustion products. While it is probably true that marijuana smoke is less carcinogenic than tobacco smoke, it has the same potential for respiratory irritation and deposits four times as much tar into the lungs as a typical cigarette, mainly because marijuana is usually smoked unfiltered. However, occasional marijuana use appears not to have a significant effect on lung function up to seven joint-years of lifetime exposure. (I chuckled when I read that term; it means one joint a day for seven years or one joint a week for 49 years). Of course, this hardly compares to a typical tobacco smoker, who smokes anywhere from a half pack to two packs a day (10-40 cigarettes), and those using medicinal marijuana can be expected to be smoking at least a couple of times a day. Medical cannabis advocates even basically admit that this is true.
In any case, if one were going to decide on a drug delivery device for cannabinoids, one could hardly design a worse device than burning the leaf and inhaling the gases, where the active drug is just one of hundreds of products of combustion, all loaded with particulate matter and tar. Sure, toking one joint a day probably doesn’t do appreciable lung damage in the intermediate term, but smoking one cigarette a day probably doesn’t either. In the case of glaucoma patients, a condition for which there is some evidence of efficacy, it’s been noted that patients would have to be toking up several times a day:
Since at least 2009, for instance, the American Glaucoma Society has said publicly that marijuana is an impractical way to treat glaucoma. While it does lower intraocular eye pressure, it works only for up to four hours, so patients would need to take it even in the middle of the night to achieve consistent reductions in pressure. Once-a-day eye drops work more predictably.
Yet glaucoma qualifies for treatment with medical marijuana in more than a dozen states, and is included in pending legislation in Ohio and Pennsylvania. At one point, it appeared in New York’s legislation, too.
And:
What’s more, for some of the ailments, such as glaucoma, patients would have to toke up every three to four hours day and night to maintain therapeutic levels in the bloodstream or tissues. Routinely consuming that much weed would be incapacitating.
Clearly, even if marijuana is efficacious for some conditions, there are serious drawbacks to burning the plant and inhaling the smoke as a drug delivery system. Other problems exist, not the least of which are the psychoactive effects of THC, which cause much of the “high” that pot smoking produces. To paraphrase one of the ophthalmologists in the NYT, his 60-year-old patients with glaucoma don’t want to be stoned all the time to get the beneficial effect of medical marijuana. The high is a particular problem for children, but none of this has prevented parents with autistic children from claiming that pot can treat autism, complete with seemingly-heartwarming anecdotes. One can imagine the temptation to simply keep the child toking until he becomes mellow and more “manageable.”
Of course, medical marijuana being in essence herbalism, with the same claims for efficacy of the “whole plant” due to synergy of its ingredients and the same attitude that “natural is better,” it’s not surprising that the same problems exist that are routinely observed for any herb sold for medicinal purposes. These problems include as inconsistent potency and purity, adulteration with contaminants—or even questions of whether the plant being sold is actually what is being claimed. Indeed, a fascinating story that sounds very familiar to those of us who have been paying attention to adulterated herbs and supplements was published a month ago in The Seattle Times:
Tonani, 38, decided several years ago to try pot. And it has worked for her, she said, especially strains low in the psychedelic chemical THC and high in the non-psychoactive ingredient cannabidiol, known as CBD.
As a medical-marijuana patient, Tonani knows it can be hard to find the rare strains that don’t make you high — and it can be even harder to get the same kind of pot consistently.
Testing shows that some marijuana strains are not what they purport to be in name, chemical content and genetics. This is particularly concerning for patients seeking pot low in intoxicants and high in pain-relief or other therapeutic qualities.
One strain widely known for its high-CBD and popular among medical-marijuana patients is called Harlequin. But when Tonani and a leading Seattle pot-testing lab analyzed 22 samples of Harlequin from various growers and dispensaries, five of them were high in THC and had virtually no CBD, which means people trying to take medicine were just getting high instead.
Again, this is a very common problem with herbal medicines, and cannabis, when smoked or ingested as the plant, is an herbal medicine.
Medical cannabis: Politics versus science
There’s no doubt that what is driving the legalization of medical marijuana in so many states has far more to do with politics than with science. Right now, for all but a handful of conditions, the evidence is slim to nonexistent that cannabis has any use as a medicine, and those conditions, such as CINV and chronic pain, can often be treated more reliably with purified or synthesized active components. Moreover, for one condition for which there is reasonably good evidence for the efficacy of cannabis and/or cannabinoids, namely chronic pain, politicians are reluctant to approve medical marijuana, as described in the recent NYT article:
Often state legislators have been motivated not just by constituents in distress, but also by the desire to restrict access to limited patient populations so that legal marijuana does not become widely available as a recreational drug in their states.
For example, while there is research suggesting that marijuana alleviates certain kinds of chronic pain, Mr. Lang noted, legislators in Illinois were reluctant to legalize its use in such a broad patient population. The state’s list of qualifying conditions is lengthy partly because lawmakers tried instead to specify a number of diagnoses that result in pain, some quite rare.
“I’ll bet there are hundreds of conditions that cause pain, and now 30 are listed,” Karen O’Keefe, director of state policies at the Marijuana Policy Project, said of Illinois’s legislation.
So, for one indication for which there is reasonably good evidence for the use of cannabis, legislators in Illinois were reluctant to approve its use, while approving its use for a lot of indications for which there is no evidence to support them. Clearly, this is a policy area that cries out for better science, given how legislators are being swayed by anecdotes that do not demonstrate that cannabis is effective and stories of “persecution” for growing medical marijuana, rather than by well-designed randomized clinical trials. Add to that the conflict with currently existing federal law, which outlaws cannabis as a schedule I drug, and the political situation is a mess, making doing research to find out for what indications cannabinoids have efficacy much more difficult. Antidrug zealots hugely exaggerate the danger of pot smoking, while pro-medical marijuana zealots claim that “cannabis cures cancer.” (It doesn’t, as I will discuss in the next installment.)
Moreover, THC can have biphasic activity:
THC has what doctors and researchers know as biphasic activity. “At low doses it has certain effects, and at high doses it has opposite effects,” Dr. ElSohly explains. “Somebody using to get high at the right dose will be calm, happy, getting the munchies, and all of that,” Dr. ElSohly says. Someone using at the right dose could see medicinal benefits, too. But take in too much THC, and you can become irritable, even psychotic. “There are more emergency room admissions today than ever because of marijuana use,” Dr. ElSohly says. “That’s simply because of the psychoactive side effects of the high THC content that the public uses.”
This makes standardization and getting the dose right more important for medical cannabis than for most other drugs, which is why I’m not enamored of smoking pot as a THC/CBD delivery system. At the risk of being too personal and “anecdotal,” I couldn’t smoke pot if I wanted to, for recreational or medicinal uses, whatever my feelings about its legalization. I can’t smoke cigarettes, either, and have never tried either pot or cigarettes. The reason is simple. Inhaling just secondhand smoke sends me into fits of coughing—and has since I was a child. Inhaling smoke directly into my lungs has been and still is more or less unthinkable. And I’d bet I’m not alone, either.
My personal sensitivities aside (which are obviously not shared by most people), I see two critical unaddressed questions with respect to cannabis. The first issue is standardization. I’m sorry, herbalists and pot smokers, but smoking a dried plant just isn’t it, particularly given the relatively low doses of active compound needed for optimal effects. That means pharmaceutical-grade material. If cannabis is a therapeutic drug, it should be treated like every other therapeutic drug and be subject to clinical trials. The second issue is comparative effectiveness research. It’s not enough just to say cannabis (or whatever cannabinoid drug or derivative you might wish to use) is “efficacious” against this disease or this condition. We need to know how efficacious it is compared to the existing standard of care. In most cases, even for indications for which there is evidence of efficacy, the existing evidence base suggests that cannabis is less effective than existing treatments, with the possible exception of its use as an antiemetic. Yet none of this sways the zealots, just as similar evidence with respect to other herbs doesn’t sway believers in herbalism. Meanwhile, medical cannabis is rapidly becoming big business.
That’s because cannabis is the new herbalism. With relatively few exceptions, it’s about belief first, and then trying to get the science to to support its magical properties.

235 replies on “Medical marijuana and the new herbalism, part 1”
Orac, what a breath of fresh air! Well written, accurate, and accurately representing the state of research. You continue to impress me writing on an area that I usually cringe about.
I’m a bit frustrated by other medical sources and skeptics who deal with cannabis from a pro-legalization perspective.
Here’s my position:
1. Medical uses should have no bearing on whether a drug can be used for recreation. Codeine makes a great cough syrup, but I don’t think that means it should be legal to sell at the local gas station or 7-11. Likewise, hydrocodone is an indispensable drug for pain management, but legalizing its use for recreation makes no sense. If cannabis has medical applications, let it be treated the way we treat prescription drugs. We shouldn’t mix and match and encourage self-medication.
2. Tobacco and alcohol are not legal because they are safe. Let’s keep reminding ourselves that, combined, these two legal drugs are involved in causing more preventable deaths than any other single factor. They are only legal because we lack the political will to make them illegal.
To me, this is no different than saying we should release any serial murderer who killed fewer people than Jack the Ripper, because that guy never served any jail time, so why should anyone else? Let each drug stand or fall on the merits and harms shown by research.
I’ll stop there. This is a sore issue for me. I’m a little frustrated with a lack of clear, rational thought on the issues around physicians encouraging the legalization of a recreational party drug. I appreciate this well-written article.
Yes, I can see why a substance well known for producing cottonmouth would be a natural therapeutic choice here.
On the antiemetic front, I don’t know how long it would typically be prescribed for, but longer term use carries some risk of the peculiar cannabinoid hyperemesis syndrome.
You never smoked marijuana? Well your GPA was probably a lot better than mine.
The irony for me was that when I was prescribed Marinol for weight gain I hated it. Threw all the pills out. When you are taking a lot of meds as I was at the time, the last thing you want is another med with central nervous system side effects. IMHO. Plus I never really was one for getting the munchies – but I sure loved music & art! Sometimes I kind of miss it when I go to concerts now. sigh.
And because it’s my job to basically pick from the bottom of the barrel in science blogging…
…Well, there’s this guy.
http://phoenixtears.ca/
Specifically, his “how it works and what it does” page, where he writes, I shit you not:
“Many people who have taken the oil have stated that they thought it to be the fountain of youth. From my own experiences with the oil I believe this to be true.”
I’m as much a fan of the odd johnny as the next fella, but seriously, backing up attempts to legalize a drug with dishonest science is just fucking dumb.
@c0nc0rdance
I seem to recall that alcohol had indeed been made illegal in the US, circa 1930?
Did it work SO AMAZINGLY well?
Prohibitioning aside, I am in agreement with 100% of what Orac said. Interesting possibilities indeed, but blown out of proportion.
I’ve been a long time lurker, finally commenting!
This was a great post. My nephew has Dravet, and my mother was talking a few years ago about taking him to CO for cannabis oil. I think I talked her out of it based upon dosage/quality arguments learned here from our esteemed host (or perhaps that’s a bit egocentric-his mother may have simply nixed the idea). Been following epidiolox (sp?) for about a year now-they’ve opened up a trial in the midwest and that may be a possibility for my nephew if his neurologist agrees.
As much fun as this blog is, I rather miss some of our more “entertaining” visitors. I know Greg asked to be banned (still gives me the grins), but whatever happened to Augie, or Jen, or Sid? I know getting spanked isn’t fun, but their apparent decisions to leave us left a deficit in my daily dose of giggles.
Trying to ban stuff that can be grown (tobacco, pot, grain for alcohol) and then easily produced with household ingredients/equipment (alcohol, there might be more but I don’t know the facts there) is an enormous game of whack-a-mole that the moles definitely will win.
I’ve heard that synthetic pot is more dangerous than plant pot. Don’t know if it’s the usual anti-drug scaremongers, or if the unintended consequences have actually become a bigger problem than weed let well enough alone.
Disclosure: never been a user and I only say that to make clear I have no agenda other than wanting to see the end of the likes of the DEA and military involvement in stuff that is not in its charter. Way too much resources involved in chasing pot for no benefit and likely much harm.
I believe there’s a place in the world for healthful, non-medicine products. For example, my wife will drink a cup of coffee when she’s having asthma rather than jumping right to the emergency inhaler. Could that instead be an FDA regulated pure caffeine powder? Sure. But coffee works.
So certainly claims of Cancer and Glaucoma cures need to be treated as medicine and carefully studied with specifically extracted compounds. But if a cancer patient finds a pot brownie helps their nausea or appetite? Great! Even if it were just placebo, that’s still a good thing.
I think that’s where this confusion of marijuana and medicine comes in. It’s not that it needs to be a medicinal appetite stimulant, it’s that if a very sick person finds help in it, why not just let them consume it? Not as medicine, but as something fairly benign that helps them feel better. But since marijuana is illegal it needs to be couched in the terminology of medication to get a legal exemption for the individual. Which as you point out confuses the issue of what a medicine is.
I’ll reiterate some other commenters, this was a remarkably clear-headed piece on this topic.
I’ve heard that synthetic pot is more dangerous than plant pot.
I’m willing to believe that synthetic pot is more potent than plant pot. It’s easier to adjust the THC content of synthetic pot to what you want, and I would think[1] that for most users higher is better. Whether that’s more dangerous depends on your perspective. A user who is used to plant pot could be overwhelmed by a similar dose of the more potent synthetic pot. But with synthetic pot, at least for medicinal purposes, you know how much you need, unlike with plant pot where the potency can vary from negligible (ditch weed) to quite strong. The latter comes up with other herbal medicines: in synthetic form you know how much you are getting, whereas with grown product you are never sure.
[1]Not that I have any personal experience on this topic….
I think B&F may have been referring to compounds such as JWH-018, which are full agonists of the cannabinoid receptors.
I mostly agree with this but I think one of the reasons patients prefer to smoke is that it’s actually easier to control the dose then with a pill, such as Marinol. I think most people who use it for medicinal purposes probably just take a puff or two at a time, wait to gauge their reaction and then smoke a little more if needed. I had a friend with abdominal cancer who after surgery and chemotherapy decided to quit treatment. He said narcotic pain medicine made his pain worse because it constipated him. Pot and hot baths in his jacuzzi were the only things that helped. And anyone who smokes a whole joint of todays’ marijuana is hard core. I haven’t smoked in years, but once they came up with the powerful strains, i found one or two tokes was all I could handle. As far as nausea meds, at least some of them have potential extrapyramidal side effects. I believe they black boxed Reglan for this. And I think compazine and phenergan also have that potential. Don’t know about Zofran, though.
T: You’re a bit off. the ban started in 1920, went through 1932 when it was finally repealed.
Some of the stuff sold as “synthetic pot” may not be the active ingredients in pot just derived synthetically. Some of the so-called “bath salts” are sold as synthetic pot.
How much they are really like the compounds in pot I don’t know (as last I heard on the news they are whichever designer drugs hasn’t been banned yet and as one gets banned a new compound gets put in the products). I assume they call the whatever they cooked up synthetic pot because people don’t think of pot as dangerous so it makes them more likely to buy it than whatever chemical they put in it this month.
I really doubt that MDPV and friends are smokable.
One thing smoking ones pain drugs would do would be to normalise it, to make it social. Everyone i know on opiates, and these are prescribed ones, is under constant censure and judgement from others, often their family, for taking them. Even when the pills are all that allows them to function.
Turning pain relief into a fag break would help.
note for merkins…fag in English means cigarette. If fag break sounds odd to you think how the reference to a johnny sounded to me upthread. As a johnny is a prophylactic…
Well, those are “jimmies” here, but I think that’s mostly Black slang (as is “squares” for cigarettes, which seems to be falling out of use, although I still say it, sometimes to the surprise of the hearers; “that’s jail talk, man”).
I thought that jimmies were sprinkles. Live and learn.
MO’B, I didn’t know you were a fellow Massachusettsian!
@Antaeus Feldspar – I lived in Mass for 4 years ago some time back. I learned the difference between a frappe and a milkshake and what a tonic is.
Google patent US 6630507 Heard of this?
Boy its hard to stay away from this place 🙂
I have personal experience and yes what I am going to do is provide an anecdote so keep that in mind.
I have cancer and have had chemotherapy for it. I did take cannabis to try and put a lid on my nausea. While it helped a bit it had no where near the effect that the anti emetics that were given to me did. Compared to Aprepitant it was pretty useless.
As for analgesia it works but for me it is no better than paracetamol. Codeine has a much greater effect. I know this as I also have Rheumatoid Arthritis. It is also claimed that cannabis is an effective anti inflammatory agent. Well compared to Celecoxib it really is ineffectual. As for modifying RA’s effect on the joints compared to methotrexate it is useless.
I am a member of a cancer support forum. We regularly get people on board who claim that cannabis is protective against cancer. I really find that hard to believe as I personally know stoners who ended up with cancer. What I really want to see is a large scale study that investigates the incidence of cancer in people who regularly use Cannabis for recreational purposes. I could then point to the study and shut them up. Mind you it is a support forum so any argument is short lived. it would be nice though to have some evidence to educate the clueless.
Someone commented on political will to make things illegal. Well we tried that in the 1920’s with alcohol and it was an unmitigated failure. Making things illegal does nothing but empower criminal organizations and turn decent citizens into criminals. We have had a war on drugs now for decades and nothing has happened except tens of thousands have died in drug related violence and the use of illegal drugs has skyrocketed. I applaud those countries and those states in the US who have made it legal for recreational purposes. Make them legal, tax them, then use the money in public health campaigns. It has worked for tobacco and it will work for cannabis, or any other illegal drug for that matter. In the US and Australia we have managed to get smoking rates down from over half of all men down to around 15%. We never had to resort to banning it and to do so I think would be counter productive.
So in passing if anyone can point to a large study showing cancer incidence in heavy cannabis users I would be most appreciative.
Cheers
I challenge you to a public debate, you paid-for, antiscientific shill! Continue to help the Prohibitches withhold God’s healing plant and reap the karmic consequences…Repeal – Amnesty – Reparations! We will settle for nothing less. Respect existence or expect resistance 😉
Did you even bother to read the article, not-so-Bright?
AdamG, did you not notice the winking emoticon at the end of Drew Bright’s comment? Drew is Poeing.
Yup. But if your point was “there must be something to it, or they’d never have gotten a patent granted” then you’re quite mistaken. Patents have been granted for many things that don’t do what they’re claimed to – for perpetual motion machines, or for infinite data compression methods (which, believe it or not, are actually more impossible than perpetual motion.)
I’m still convinced that a really good compression method using deduplication and zero suppression should be able to take any data and compress it to a single bit.
G—gle patented cannabis? Oh, wait…. Your point is that CB1/CB2 affinity is wholly unnecessary?
Who owns controls that patent?
@blinkyeb – are you here to make some point or to play 20 questions? If you’d like to make a point, please state it. Thanks.
You didn’t understand that whole receptor business, did you? Do you surmise that the best acute treatment for stroke is trying to figure out how to get a joint into the patient ASAP?
MOB “I’m still convinced that a really good compression method using deduplication and zero suppression should be able to take any data and compress it to a single bit.”
Yes, but it only works reliably for political speech and marketing copy.
There are any number of techniques that would suffice for compression to a single bit. None of them, however, have corresponding de-compression techniques.
rs – I’ve heard of a two-bit politician, so it’s within reach.
Bill Price – that’s where it all falls down, of course, unless one has a quantum computer.
that’s where it all falls down, of course, unless one has a quantum computer.
In other words, Deepitypak can do it, but not us regular folk?
You’re giving me flashbacks to Barnsley’s “fractal compression” algorithms from the 1980s.
Oh I googled around before commenting, and sadly Mr. Bright is not a Poe.
If you were wrong about marijuana 20 years ago, maybe it’s also possible that you’re wrong now about some of the things you describe as “woo.”
>”Inhaling just secondhand smoke sends me into fits of coughing—and has since I was a child. Inhaling smoke directly into my lungs has been and still is more or less unthinkable. And I’d bet I’m not alone, either.”
Actually, unless you have allergies, asthma or some kind of respiratory condition, that’s VERY unusual. Most people can inhale smoke no problem.
jacksvsworld, a quick question:
Did smoking pot make you stupid, or were you stupid before you started to smoke it?
I ask this because I spent two hours a couple of days ago waiting for old Honda that was parked in my driveway to be removed by the police. Not just blocking the driveway, but backed to being not far from the garage. In the back seat was a bong, and the police officer told me it was a stolen car.
What kind of intelligent person steals a old small car and then backs it up a driveway, while leaving their stuff in the back? Needless to say, I am not impressed with that person’s cognitive function.
By the way I only smoked pot for a week in college because it gave me annoying “itchies”, and I did not like that it made me stupid. I also have a real verifiable allergy to nicotine, so I don’t like being around tobacco smoke. Or wood smoke. Or any other airborne particulates.
‘Most people can inhale smoke no problem.”
Really? Provide the PubMed indexed studies from qualified reputable researchers to prove otherwise. While we have had several in our family die from tobacco smoke inhalation (including a 42 year old who lost his jaw to cancer), I will only use the sixty plus years of epidemiological studies showing tobacco smoking is deadly.
Since pot smoking has only become legal in the past couple years in a few places, I predict there will be similar results from studies in the next forty years for marijuana.
By the way, I voted to legalize marijuana smoking in my state. I really don’t care what chemicals you inhale, I just don’t want doing it in my air space. Committing acts like stealing a car and abandoning it in someone’s driveway may or may not be the affect of smoking pot.
It just does not reflect well on the users. So please clean up that image, unlike this city attorney. You can start by being coherent.
And it’s also possible that “jackvsworld” is actually a criminal wanted by the FBI for sordid acts of bestiality against endangered species.
It’s not enough to say something is ‘possible’.
A small amine isn’t able to act as an allergen. It would have to somehow function as a hapten, but with a protein that also ignored vitamin B3.
Narad, you’ll have to take my skin tests results with the allergist MD who explained them to me. Personally I was glad in the late 1970s to have a verifiable piece of paper to bolster my annoyance at those who wanted to smoke tobacco in my presence.
That included my dorm roommate who chose a non-smoking room thinking it would help her quit. Oh, and the folks in movie theaters, and the woman seated in the nonsmoking section of the airplane, and the woman in our office who kept telling us how we should only eat organic food while sucking on a cigarette (so glad when the company went smoke free, and surprised when some tried to hide the lit cigs under their desks).
I just hope that we can convince the ones who wish to now legally smoke marijuana can be reminded that many of really find it annoying.
By the way, not all things that cause reactions are proteins. Though sometimes things that cause dermatitis do that as a combined reaction to items in the body, like nickel reacting to perspiration.
Note I was very upset that some idiot abandoned a stolen car with a bong in on my driveway that trapped me from driving somewhere before it closed. Plus waiting two hours after calling the police for it to be removed (had to resort to “911” after spending several minutes on hellish voice mail trying the “non-emergency” parking infraction police number).
That alone illustrates one reason why I detest the idiocy of folks like jacksvsworld. Oh, and pedantic comments that may follow. So if you’ll excuse me I will still think very little of the intellectual capability of anyone who says “people can inhale smoke no problem.”
Because it doesn’t take a rocket scientist (fortunately I used to be one) to know that it is not healthy to intentionally inhale particles contained in any kind of smoke.
There are patients that have beat cancer and other llife threatening disease with cannabis, most have not let the Dr’s give them the poison called chemo, they went straight to the oil made from the plant tha heals most anything it somes in contact with. With holding this plant from people (specially the seriously ill) is inhumane. Do your homework, there are tests showing the power of this plant , I have seen brain tumors gone with no evidence of cancer left after using the medical marijuana oil and eating a clean healthy diet. The most important thing you can do for yourself is to eat right, no GMO foods, no sugars etc and educate yourself on all the things this amazing plant can and does heal!
There are patients that have beat cancer and other llife threatening disease with cannabis, most have not let the Dr’s give them the poison called chemo, they went straight to the oil made from the plant tha heals most anything it somes in contact with. With holding this plant from people (specially the seriously ill) is inhumane. Do your homework, there are tests showing the power of this plant , I have seen brain tumors gone with no evidence of cancer left after using the medical marijuana oil and eating a clean healthy diet. The most important thing you can do for yourself is to eat right, no GMO foods, no sugars etc and educate yourself on all the things this amazing plant can and does heal!
Nice prohibitionist hit piece, Orac. You lose when your already name calling with terms like quackery in the first paragraph. See, I can spot this thinly veiled pseudo science, limited hangout, feces a mile away.
Just word search “Ceramide” in the article.
Hmmm ….. it`s not there, Orac. That means your either not a very good researcher, or an author with an agenda who is suppressing some very important info some of your readers may find helpful.
In the words of Dennis Hill Bio-Chemist who cured his prostate cancer with cannabis oil.
“In every cell there is a family of interconvertible sphingolipids that specifically manage the life and death of that cell. This profile of factors is called the “Sphingolipid Rheostat.” If endogenous ceramide (a signaling metabolite of sphingosine-1-phosphate) is high, then cell death (apoptosis) is imminent. If ceramide is low, the cell is strong in its vitality.
Very simply, when THC connects to the CB1 or CB2 cannabinoid receptor site on the cancer cell, it causes an increase in ceramide synthesis which drives cell death. A normal healthy cell does not produce ceramide in the presence of THC, thus is not affected by the cannabinoid.”
Cannabis, specifically THC, kills cancer Orac. There is zero doubt. It has been demonstrated endlessly for at least the last 20+ yrs. In a legalization debate, a retired DEA agent was forced to admit they knew of some 10,000 peer reviewed articles as of 2007. Who knows how many now.
And once a majority of world citizenry know this, the for-profit-not-for-healing big cancer industry is going to a multi-billion dollar financial hit.
Is that what your protecting Orac? Or are you just uninformed?
And Dennis Hills account is just one of countless Cannabis Therapy success stories. That`s why GW Pharmaceuticals applied for some 43 patent for various aspects of the production of their multi-spectrum medicine Sativex.
Do you really think the US gov. has multiple patents thru Health and Human Services for no reason?
Then, lets get one thing straight, so to speak. Smoking cannabis is a great way to get high, but a lousy way to take advantage of the super nutrient/medicinal value the plant has. It has to be eaten, preferably as a whole plant extract.
And then lets get another thing straight. We the People do not need, nor want any more research on this plant. It is the most researched plant in human history.
What we want is complete deregulation. Just like pre-1937. The world got along just fine with cannabis as it had for some 5000 years previous, and so far Colorado and Washington have proven it will be largely the same now.
We want to be able to grow this plant for any purpose we so desire. And since it is already well established that cannabis has an astronomical LD50 rating, we don`t need the medical/pharma complex to tell us what the proper dose is. After all their products, and dosing guidelines are responsible for many thousands of deaths and injury yearly. We`ll figure it out, thanks.
Nothing less than complete deregulation will do. And we will get it.
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3770515/
“Endogenous cannabinoids regulate the de novo synthesis of ceramides, lipid-based components of the cell membrane that perform both structural and signaling functions. It is becoming increasingly obvious that ceramide functions as a physiological signaling molecule, particularly with regard to the control of apoptosis, but also growth arrest, differentiation, cell migration, and adhesion.104 As such, the role and regulation of ceramide signaling is attracting increasing attention, and ceramide now has an accepted role in the development of some cancers.105 Activation of either CB1 or CB2 in glioma cells is associated with an increase in ceramide levels leading to the activation of the extracellular signal-regulated kinase (ERK) pathway via Raf-1 activation and p38 MAPK activation.14,106 Both these pathways ultimately cause apoptosis through caspase activation and/or cell-cycle arrest.14 In breast cancer cells, the CB1 antagonist SR141716 inhibited cell proliferation through the effects of ERK1/2 colocalized inside membrane lipid rafts/caveloae.59 Such rafts play a critical role in the growth and metastasis of breast tumors.107,108 A final component of the ERK pathway, p53, plays a crucial role in switching between cell-cycle arrest and apoptosis.109 In cultured cortical neurons, Δ9-THC activated p53 via the CB1 receptor, thereby activating the apoptotic cascade involving B-cell lymphoma (Bcl)-2 and Bcl-2-associated X protein, suggesting that the cannabinoid pathway ultimately causes cellular death via apoptosis.110”
@Danman
You missed the conclusion from that paper you linked to, in which the authors say that we cannot move forward with cannabinoids as a cancer treatment without more research to resolve disagreements in the litarature. To wit:
Marijuana Study Shows No Lung Cancer Risk
“Dr. Donald Tashkin UCLA Geffen School of Medicine Pt 2 of 2. Conclusion of 2 part interview with the famous research doctor from UCLA Geffen School of Medicine. Pulmonary research on use of marijuana and interaction with the lungs was funded by the Federal Government to prove that lung cancer is caused by smoking marijuana, however the results proved cannabis does not cause lung cancer.”
Citation via YouTube? How quaint.
PubMed is your friend and there are many more recent studies showing the deleterious effects on pulmonary functions and the risk for lung cancer:
http://www.ncbi.nlm.nih.gov/pubmed/?term=cannabis+and+lung+cancer+risk
Todd W ….. point taken. And absolutely, they can resolve the scientific differences til their blue in the face. No problem. After complete deregulation.
Because regardless of the exact science and bio-mechanisms involved, there is, and never was any credible justification for the prohibition of cannabis in the first place. That`s just a fact.
Prohibition is nothing more than the DEA enforced monopoly on what “We the People” can, and cannot, use for food, medicine, or to get high with if we so choose.
And fine, let the pharmaceutical industry have at it. If they want to make a cannabinoid profile certified whole plant treatment for any number of diseases, ala the GW Pharma approach, for say $2000 for a cancer treatment regime, that`s reasonable. Insurance could cover 75% and the patient could shell out $500 out of pocket. That would be a whole lot better than losing all your savings, your job, your house, everything, after a long, expensive, brutally torturous, and ineffective conventional treatment regime.
And some people will prefer that approach. But many of the rest of us are well acquainted with the various ways cannabis can be consumed as medicine and will continue to make and use it as we please, legal or not.
And unless the authorities_that_shouldn`t_be shut down the internet, it`s a genie that cannot be put back in the bottle.
“And once a majority of world citizenry know this, the for-profit-not-for-healing big cancer industry is going to a multi-billion dollar financial hit.”
This has been said about Rife machine treatments, laetrile, Hoxsey therapy, essiac and many other forms of cancer quackery.
“We want to be able to grow this plant for any purpose we so desire.”
Obviously. And I don’t want to stand in the way of your legal high.
Just stop using cancer patients as a tool to get what you want.
Come on lilady, really?
There has been 50+ years of 100`s of millions of people smoking cannabis on a daily basis around the world. If the was any real threat of cancer, lung or otherwise, from cannabis, smoked, or eaten, the US Gov. would have been able to DEFINITIVELY prove it. Period.
It only took the gov, about 20 years to fully prove tobacco causes cancer, that despite the huge lobbying power of the cigarette industry.
They have not been able to prove a link to lung cancer because it does not exist.
Dangerous Bacon …. the QUACKERY is highly reactive toxins, and x-radiation for cancer….. unless you want to kill EVERYTHING, which it does.
Considering cannabis is mostly smoked in combination with tobacco and tobacco smoking raises the risks of getting lungcancer, it isn’t that weird that there might not be much research to prove cannabis raises the cancer risks. It is proven to raise the risks of getting a psychosis.
And don`t get me wrong Bacon ….. your welcome to go for the modern QUACKERY. Enjoy.
Renate ….
A) Cannabis is not usually smoked with tobacco.
B) That would not be a cannabis lung cancer study. That would be a cannabis/tobacco cancer study.
C) If you sprinkle arsenic on your corn flakes you no longer have a healthy breakfast.
Just keep the cannabis away from me. It stinks to high heaven.
Now that it is legal in my state the idiots think they can smoke it anywhere. Even though the law explicitly states it must be done in private. And why do the dolts think that those of us who dislike smelling tobacco smoke think we would be okay dokay about sniffing the stench of their smoke?
Oh, and Danman, because the consumption of cannabis smoke was previously illegal, there could be no studies on lung issues. I suspect that the users may not be quite truthful on their habits to their treating oncologist.
After ten to twenty years of legal usage, then you might get a large enough sample to do an epidemiological study.
And hopefully by then they will realize that toking is as socially acceptable as tobacco smoking in public, so the stench will not invade my breathing space.
Citations needed, Stacey.
You’ll have to excuse me if I wouldn’t touch some half-assed butane extraction with a windowpole.
(Fun fact.)
I assume by this you’re referring to current standard-of-care radiation therapy and chemotherapy. I’ll point out that there exists a large body of evidence, including large scale clinical studies, demonstrating the efficacy of these as treatments for cancer, alone or as an adjunct to other medical interventions such as surgery.
As no such body of evidence exists which suggests cannabis is effective at treating cancers, it’s pretty clear who’s promoting quackery here.
There is an official study showing cannabis extracts treated terminal acute lymphoblastic leukemia in the November 2013 Case Reports in Oncology issue (http://ncbi.nlm.nih.gov/pmc/articles/PMC3901602). It actually ruled out spontaneous remission and chemotherapy as potential causes.
I also suggest people check out CannabisExtractReport.com, which includes medical documentation of numerous terminal cancer patients in remission. I’m not going to argue this here, you can see the evidence for yourself and understand how real this is. Doctors, corporations, dispensaries, and small teams are all reporting these remarkable anti-cancer effects. See how deep the evidence is and judge it for yourself.
THC is a CB1 agonist, Einstein.
Apparently, you don’t get out of “The US-of-A” much.
Really? Everyone I knew who smoked cannabis, mixed it with tobacco. I”ve smoked it myself. Well, of course, there is spacecake, but that you don’t smoke and I’ve never eaten it, not felt the need to.
Danman,
Cannabis is not some wonder drug, much as you’d like it to be. As noted above, it does promote psychosis in those predisposed toward it. Also, smoked cannabis, as with any smoked item, increases the risk of diseases of the lung, including cancer.
As to your study showing cancer risk, as also noted already, how exactly were these studies to be done when it was illegal? The study would also need to exclude those who smoke or have smoked tobacco products, since that would confound the results.
Finally, the whole plant would likely not be a particularly good medicinal treatment, since it has not only those compounds that would act as a treatment, but lots of other contaminants that would produce undesired side effects. I agree that it should be studied to see what active compounds can have a medicinal effect, but it is not a panacea.
“In breast cancer cells, the CB1 antagonist SR141716 inhibited cell proliferation through the effects of ERK1/2 colocalized inside membrane lipid rafts/caveloae.”
THC is a CB1 agonist, Einstein.
———————————————-
No it`s actually CB1/2
, Forest ….
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2931553/table/tbl2/
Citation needed.
After all, if it has actually been established as fact for 20 years that cannabis cures cancer there must by now be a substantial number of Phase III clinical trials attesting to its efficacy.
I know that. Please try to understand what you’re cutting and pasting first before misunderstanding my reply. Jeezums, it was about as dumbed-down as possible already.
In fairness, this is a more complicated question.
Sigh. Anecdotes of cancer cures are not good evidence. I suggest that “Danman” and Justin search this blog for “Stanislaw Burzynski” and “success story” or search it for “cancer testimonial.” I’ve published more posts than I can remember describing how “cancer cure testimonials” almost certainly are not good evidence of a real cancer cure, coming, as they nearly all do, from misunderstandings about cancer.
A brief skim of Justin’s case report does not convince me there’s compelling dose-response data, contrary to the claim in the report. Also contrary to the claim in the report, spontaneous remission was not ruled out.
Daman, there’s a fairly large body of literature regarding the potential of targeting ERK/MAPK pathways to treat cancers, and several large pharamceutical companies have ongoing research programs attemtping to develop novel inhibitors, which have identifed a number of small molecule inhibitors representing different classes.
What suggests cannabinoids as a class are likely to perform as well or better than all other ERK inhibitors?
Come on people, stop with the idiotic paradigms …… Just because it`s illegal doesn`t mean a definitive link to cancer could not be established. That`s just silly talk.
I repeat;
There has been 50+ years of 100`s of millions of people smoking cannabis on a daily basis around the world. If the was any real threat of cancer, lung or otherwise, from cannabis, smoked, or eaten, the US Gov. would have been able to DEFINITIVELY prove it. Period.
It only took the gov, about 20 years to fully prove tobacco causes cancer, that despite the huge lobbying power of the cigarette industry.
They have not been able to prove a link to lung cancer because it does not exist.
Allow me to clarify SR141716 for you, Danman: For the grossly simplified level of discussion you apparently require, it’s not all that dissimilar to a cannabis version of naloxone.
JGC …… I think you don`t get it.
Most advocates DON`T care about any more studies. The plant has been researched up one side and down the other. There was never any credible justification for the prohibition of cannabis in the first place, and there is sure no reason today.
Study it for another 80 years. I don`t care. But not under a federal ban.
And if your serious about research, a climate of deregulation is far more conducive than under a restrictive prohibition.
BTW, Daman, re: Dennis Hill? I’ll note that he was diagnosed with localized prostate cancer (Stage III–cancer local to the prostate and tissues near the prostate) around 2010. It’s hardly surprising that he’s still around: for patients with localized cancers adopting watch-and-wait strategies the 5 year survival rate is greater than 90%–there’s no reason to attribute his success to the cannabis oil (especially since his case represents an n of one.)
“Most advocates DON`T care about [evidence].”
Fixed that for you, and believe me–I get that.
I get the feeling that neither Danman nor Justin will like part 2 of this series when I get around to it…
Rimonabant, Homer ….. a very bad diet drug.
Does that mean that, in your opinion, the term “medical marijuana” is a farce? That the whole attempt to allow doctors to prescribe it and pharmacies/dispensaries to sell it has no provable medical justification? That really, the only reason that people advocate for “medical marijuana” is because they see it as either a stepping stone on the path to full legalization of recreational marijuana or as a path to de facto legalized recreational marijuana, with sufficiently compliant doctors writing scrips?
Is that what you’re saying?
Justin: “See how deep the evidence is and judge it for yourself.”
_Something_ is piled high and deep and that’s for sure.
“Official”? Anyway, given the prompt death of the patient (blamed on chemotherapy, of course), it doesn’t exactly seem to have been a promising therapeutic regime.
The authors do, however, suggest that Rick Simpson has been practicing medicine without a license.
JGG said; “It’s hardly surprising that he’s still around: for patients with localized cancers adopting watch-and-wait strategies the 5 year survival rate is greater than 90%–there’s no reason to attribute his success to the cannabis oil.”
It wasn`t a watch and weight situation. His medical people had him setup for chemo. I think they gave him 6 months without treatment.
You think that just happens? Spontaneous remission, with terminal cancer?
Not “no change in 3-4 years” but total remission in less than a year.
Come on stop pretending here, people.
You are the one that invoked it as an example of how cannabis cures breast cancer, moron.
Um, well yeah:
http://www.ncbi.nlm.nih.gov/pubmed/2206563
Honestly, go do some actual research before commenting on stuff you clearly don’t understand.
“You think that just happens? Spontaneous remission, with terminal cancer?”
Yes. All.The.Time. Now chances are that it happens to you, personally, quite low. Probability it will happen to someone, quite high.
@Narad
Oh, I agree that the effects of smoked products on the lungs is complex, in terms of the degree of impact, but from what I understand, the effects of inhaling smoke from combusted items acts as an irritant on lung tissue, thereby increasing the risk of lung-oriented diseases compared to those who do not inhale that smoke.
@Danman
Or they just haven’t spent the money to examine the question to the degree necessary. From studies that have been done, we actually don’t know what the dose threshold is for cancer risk. We do know that it does have a negative impact on airflow. (see, e.g., http://www.ncbi.nlm.nih.gov/pubmed/24384575) Certainly the risk of lung cancer is plausible.
Thankfully, physicians and researchers do care about more studies so they can find out what it does and does not work for, at what doses, and what the side effects are.
Mephistopheles O’Brien said; …… “Does that mean that, in your opinion, the term “medical marijuana” is a farce? That the whole attempt to allow doctors to prescribe it and pharmacies/dispensaries to sell it has no provable medical justification?”
No doctors do not have the right to prescribe cannabis, any more than they have a right to prescribe carrots.
It`s a nutrient. And as a matter of fact if it`s not decarbed, it has no psycho-activity at all.
Now if they want to just recommend cannabis, that`s fine.
It can, but more likely what happened is that the doctors were mistaken about the prognosis. That happens all the time.
Can you define ‘nutrient,’ danman?
However, Danman is quite wrong when he says that spontaneous regression of advanced cancer never occurs:
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2946665/
And:
http://www.ncbi.nlm.nih.gov/pubmed/24616911
AdamG said; …..”Can you define ‘nutrient,’ danman?”
No Adam, but you can. Just by a simple Google search.
Good luck, let us know what you find ….
You’re cute! According to M-W, a nutrient is
Now, can you please describe how cannabis fits this definition? do people need it to live and grow?
ROTFLMAO!!! …….Oh yes, I`m sure your doctor mentions Spontaneous Remission in the treatment plan all the time …..
Well Mr Smith, I`m sorry but the bone marrow killing chemo didn`t work, the radiation failed, our experimental treatment was a flop …… But there`s always Spontaneous Remission!!! Good luck!!!
Good work Adam!!!
You`re one step closer to your answer, Grasshopper …..
For your next assignment find out how much protein is in a single tablespoon of cannabis seeds ……
Oh, for G-d’s sake. You’ve already demonstrated that you think anything that mentions an endocannabinoid receptor is an automatic recommendation for smoking grass, even if it says the opposite (leaving aside any issue of getting it to the target in the first place).
Do yourself a favor and don’t bother with aimless digressions into decarboxylation of THC acid (which might be interesting in and of itself, although I’m not going looking), particularly given that you’re technically even wrong about this if one considers sedation a “psychoactive” effect.
The state of Illinois would disagree with that. Some states use the term “recommend” rather than “prescribe”, prohibiting sales to persons without a doctor’s recommendation. This provides an equivalent control to prescription, and I see minimal difference but you may believe otherwise.
Regardless, can I assume you agree with my basic conclusion – that backers of “medical marijuana” only really care about using that to gain effective legalization of recreational marijuana?
“Of course there’s protein in cannabis, cupcake. PROTEIN is a nutrient. Cannabis is not. See the distinction?”
No. Explain.
Just out of curiosity, what part of “The US-of-A” systematically and bizarrely eschews apostrophes?
Mephistopheles O’Brien … do you have a learning disability?
Did you read any of my previous comments?
Here I`ll put it in caps so you can see it clearly;
THERE WAS NEVER A JUSTIFICATION FOR PROHIBITION IN 1937. AND THERE IS UNEQUIVOCALLY NO JUSTIFICATION TODAY.
I have no regard for any state, nor the federal ban. I, like millions around the world are going to do and use this plant however we choose, and for whatever we choose. Legal or not.
Is that clear?
“It”? You’ve been running away from failed assertions willy-nilly. If you just had a claim that it is an illegitimate function of a state (construed broadly) to regulate The Herb, you could have said that and been done with it rather than making a sustained ass out of yourself.
Egad the stupid it burns.
Saying cannibis is a nutrient is a bit like saying my chair is a carbon atom.
There are nutrients in a cannibis plant (which you absorb by eating the plant, not smoking it). Macronutrients like protein and micronutrients like minerals and vitamins.
But just like my chair has a lot of things in it that are not carbon atoms any plant has a lot of things in it that are not nutrients.
You might say cannibis is nutritious as it has nutrients in it (assuming it has as much as any vegetable) but it is not a nutrient. Certainly not a single individual nutrients.
Although how many macronutrients and vitamins you get from a very tiny amount may not make a big difference in your diet. After all people don’t normally eat cups of the leaves, nor do they usually eat the seeds to get high, even though seeds tend to be nutrient dense to help the new baby plant grow.
Chris: Just keep the cannabis away from me. It stinks to high heaven.
Seconded.
Danman: No doctors do not have the right to prescribe cannabis, any more than they have a right to prescribe carrots.
Fun fact, the ‘carrots lead to better eyesight’ theory was actually just government propaganda. The British needed to explain how their planes kept shooting German planes down at night. While cannabis may have some medical benefits (improving appetite in certain patients) I doubt it’s a cure-all.
Danman’s whole point seems to be that he likes cannabis. Therefore all positive claims about it are true, and all negative ones are false. Everyone who disagrees with any claim he makes automatically agrees with everything that is incorrect.
Egad the stupid it burns ….. yes it does KayMarie.
Like not being able to spell CANNABIS correctly then carrying on as though you know what your talking about.
madder said; …… “Danman’s whole point seems to be that he likes cannabis”
Hot Damn that`s FN brilliant, madder.
Your an FN genius dude …. But that`s not my point.
A spelling troll, one of the lowest of the trolls. And yes, I usually end up committing a meme when using one.
At least my elementary school science and English teachers taught me the definition of nutrient and how it differs from a whole plant. I may have missed the spell the illegal drug properly lesson, but seems you missed a few more.
It’s not much of a vegetable* as far as I can tell, for which the only sensible interpretation is the leafy portion. All the claims seem to be more random burbling about endogenous cannabinoid receptors magically inducing a flood of antioxidants. (NN, of course, has an item about juicing it; I didn’t look to see whether it was lifted from the plethora of similar ones or vice versa.)
The crude leaf is basically rank as a foodstuff; the standard extraction for cooking purposes is to dry and then hold at a simmer in an oil–water mix, which limits the temperature and at least partially washes out the water-soluble components. The end product is waxy (which screws up standard baking recipes) and still desperately in need of flavor masking.
One can natter on all day about the EFA profile of the seeds, but this has very little to do with the medical claims, aside from being inversely correlated with the price of weed on the corner.
* Other than the time that Grady made a salad out of “wild parsley” on Sanford and Son.
Ya know, you geniuses here at Scienceblogs should let those silly guys at GW Pharmaceuticals know they`re just wasting time and money on this cannabis stuff …..
http://www.faqs.org/patents/assignee/gw-pharma-limited/
I’d say that not knowing the difference between an agonist and an antagonist and “then carrying on as though you know what your [sic] talking about” is rather more damning.
Oh, Jeezums, nobody expects the Spatent Inquisition.
Needless to say, Danman didn’t look too closely at his list (which he likely blindly picked up from, oh, say, cureyourowncancer-dot-org or “Herblover’s public profile”). Why anybody is supposed to be impressed by patent applications is an open question, but let’s consider one, No. 20100317729: “New Pharmaceutical Formulation comprising Cannabidiol and Tetrahydrocannabidivarin.”
Guess what THCV is? Yup, another antagonist. Paragraph 21 is rich:
Whatever, patent the whole lot. In any event, the thing is effectively a demonstration of the irrelevance of THC per se.
I suppose I might as well wrap it up with something that has actually received a U.S. patent in this regard.
Narad said; ……. “In the applicant’s co-pending International patent application WO 2005/120478, the application describes that THCV could be used in place of THC. It has been subsequently found that this is not the case. THCV has been discovered to work as a CB1 receptor antagonist, which is completely opposite from THC which acts as a CB1 agonist.
Whatever, patent the whole lot. In any event, the thing is effectively a demonstration of the irrelevance of THC per se.”
All that means is that for cancer, strains with low THCV and THC should be used if possible. Or more likely if you have a high THC strain, the plant inherently will have less THCV.
And this is well supported by cannabis caregivers who report that no significant tumor reduction starts until they have high THC content oils/edibles for their patients.
Holy F*cking Christ, you’re stupid. No, the patent is about increasing THCV.
I can’t wait to hear how these ‘caregivers’ are measuring tumor size, let alone how and where they’re ‘reporting’ this data.
How many joints are in a lid?
You-must-be-f*cking-stupid ……… agonist is good. THC is an agonist. THC promotes receptor activity and cancer killing ceramide production.
Antagonist is bad, like Rimonabant. It inhibits receptor activity and causes side effects.
So, Narad the Savant, if you were formulating a cannabis medicine and you knew one of the naturally occurring cannabinoids in the plant (THCV) was an antagonist, possibly working against the goal of receptor activity, you would try and limit the percentage of the cannabinoid.
That was my point. I can`t make it any more simple for you.
AdamG ….. “I can’t wait to hear how these ‘caregivers’ are measuring tumor size, let alone how and where they’re ‘reporting’ this data.”
Join some of the Facebook groups. Cannabis Oil Success Stories has 36,000 members. Many cancer CO success stories. Some patient post scans, others post reports, others just post their personal accounts … unless of course people just come there to lie and make stuff up.
Then why the fυck are you repeatedly citing shіt that crucially depends on endocannabinoid receptor antagonism to try to demonstrate specific anticancer effects from fυcking blowing weed, which really does not seem to have treated your head all that fυcking well, given this collection of pratfalls?
Well i am fighting lung cancer with chemo and cannabis oil. I have been on the oil for about 3 months now, the only side effect i have is hair loss and it is on its way back. Before the oil i could not sleep, had high blood pressure and terrible nose bleeds. I don’t know if it is a cure or not but the way i see it it sure can’t hurt any…Legalize Now!!
Narad said; How many joints are in a lid?
Thirty ….. roughly half your IQ.
Not fucking talking about smoking it for cancer, Naradhole …. must be eaten to get enough THC to cause significant ceramide synthesis ….
No where on this blog did I say smoking it will cure cancer …. LIAR
Although the joke* was indirectly about you, I will congratulate you on possessing adequate cognitive function to instantly know how many grams there are in an ounce while ciphering your way there and back.
* It’s a Cheech & Chong reference.
A naive person who experience of the world has all been through a haze of marijuana smoke might well believe this false dichotomy, that either these patients are deliberately lying or everything they are saying is true.
However, someone who does not understand why a patient with a serious medical condition might post a glowing testimonial claiming that their condition is much better and they’re almost certainly not going to die of it now, and actually believe everything they’re saying is the absolute and complete truth, and it doesn’t mean that their story is the absolute and complete truth, is a very naive person indeed.
Not that I’m aware of, thanks for asking. I learn better than many, less well than most. I do have many friends with various diagnosed learning disabilities (e.g. dyslexia); I’m sure they would appreciate your question as well.
This of course is completely different from the point you appeared to have been making, which is why I asked. You brought up a number of studies and patent applications regarding the medical use of cannabis and various compounds extracted from or derived from cannabis. You also brought up a number of testimonials for how well these products worked to treat various medical conditions. However, when people said that more research was needed to prove cannabis was effective as a treatment, you stated, “Most advocates DON`T care about any more studies.” This suggests to me that either the medical benefits are proven (which they aren’t, even according to many of the papers you cited) or that the whole discussion was unrelated the actual agenda.
I recall Jesse James saying something similar.
and
Citations needed, danman. Can you provide cites for studies in validated animal models (e.g., xenotransplants in SCID mice) where feeding subjects cannabis or giving them THC via oral administration reduced tumor burden, prolonged survival, etc., compared to untreated controls?
I mean, you do have SOME actual evidence to support your claims…right?
JGC said “I mean, you do have SOME actual evidence to support your claims…right?”
I thought you`d never ask …. 🙂
This is just one. Since the DEA is aware of some 10,000 peer reviewed studies, as of 2007, I`m quite sure there are thousands of repeats since 1974. Up until recently, under prohibition, no human trials have been conducted. But I think it`s safe to assume research, when possible, would certainly have been done with mice.
http://www.alternet.org/story/9257/pot_shrinks_tumors%3B_government_knew_in_'74
“In 1974 researchers at the Medical College of Virginia, who had been funded by the National Institute of Health to find evidence that marijuana damages the immune system, found instead that THC slowed the growth of three kinds of cancer in mice — lung and breast cancer, and a virus-induced leukemia”
I don`t think they knew the bio-mechanism (ceramide induced apoptosis) But cannabis science researchers more recently are well aware of the significance of ceramide with Cannabinoid medicine;
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3770515/
“It is becoming increasingly obvious that ceramide functions as a physiological signaling molecule, particularly with regard to the control of apoptosis, but also growth arrest, differentiation, cell migration, and adhesion. Activation of either CB1 or CB2 in glioma cells is associated with an increase in ceramide levels leading to the activation of the extracellular signal-regulated kinase (ERK) pathway via Raf-1 activation and p38 MAPK activation.14,106 Both these pathways ultimately cause apoptosis through caspase activation and/or cell-cycle arrest”
And I know this absolutely to be the case, first hand. A friend of mine has Non-Hodgkins Lymphoma, diagnosed early this year. Early stage, and slow growing according their medical peeps, thankfully. The PC of course said chemo. The friend said no I`m going to try cannabis oil first. If it doesn`t help then we`ll consider chemo. The PC was at first hesitant, but reluctantly gave the nod.
I told the friend about the THC ceramide connection and how it in theory could be used to plot cancer cell death during treatment with CO. I also suggested that if your insurance will pay for it then include it with other blood work. So over a month they did 3 draws and profiles, with ceramide included. The second draw, two weeks after they started the oil showed, according to their PC “significant increases in blood ceramide levels and a sharp drop in tumor markers”. The third test showed further increases in ceramide, but not as dramatic. They`re taking a high THC grain alcohol extract mixed with coconut oil taken orally 3x daily.
You do realize, don’t you, that your anecdote proves pretty much nothing about whether cannabis oil has any efficacy against lymphoma. Ceramide is not a clinically validated biomarker for lymphoma, Hodgkins or non-Hodgkins. Moreover, tumor markers often fluctuate. Moreover, you yourself say that this is an early stage, slow growing tumor, which are exactly the sort of tumors that can appear to be having a response when they are not.
You’ll have to do better than that.
Do these people even read their own cites? For god’s sake the last line of the abstract is
“Do these people even read their own cites?”
I believe that they are passed around on their own special forums, and many just do not go back and actually read what was on the actual webpage.
I recently encountered someone who posted a link and then a quote. The problem was that the quote he posted did not match what was written on the actual link. He insisted that he did not delete any words and how could we call him out on it!
Then there are those that will read it and not understand the actual words. Much like the anti-vax folks that claim diseases were going away before the vaccine was available and their proof is a graph of mortality, not morbidity. Basic vocabulary fail.
Orac said; …… “Ceramide is not a clinically validated biomarker for lymphoma”
Duh … Why are you playing dumbass? Of course Ceramide is not a biomarker for lymphoma specifically! It`s a direct marker for apoptosis generally! Why in the f*ck are you pretending like you don`t understand how significant that is?
You REALLY can`t understand how the normal process of cell death, cancerous or not, must be a major factor in ones overall health? And that a non-toxic plant that helps the immune system identify and target unhealthy cells, even cancerous, for apoptosis, would, and in reality is, quite beneficial for a variety of maladies?
And I haven`t even gotten into CBD. Even MSM hack Sanjay Gupta is on the CDB bandwagon for epilepsy. Why do think there are so many states coming on board, with at least “CBD Only” laws? The state prohibition dominoes are going down one after another. Possibly half the US could be
either, medical, or legal after the national midterms.
THC kills cancer cells by driving ceramide synthesis and apoptosis. That is a fact. I`ve more than made my case. I`ve demonstrated that professional researchers know this to be fact, and are very interested. I`ve provided the testimony of a scientific professional and his experience curing his own prostate cancer, and very eloquently explaining the role of THC and ceramide in cannabis oil cancer treatment.
Game, Set, Match … you lose.
But you folks here at agendablog ……… oh I`m sorry, I meant scienceblog ….. can play ostrich as long you want.
What the hell do you think the ‘agenda’ actually is? I personally am completely in favor of legalization. I know it can work as I live somewhere where it’s already legal. I just believe we should take a measured approach when it comes to health claims regarding this natural product, just like I would for health claims surrounding any natural product, legal or otherwise.
AdamG said; ……. Do these people even read their own cites? For god’s sake the last line of the abstract is
“Overall, there is still a great deal of conflicting evidence around the future utility of the cannabinoids, natural or synthetic, as therapeutic agents”
Yawn …….. so what? That`s called scientific process. Every scientific theory has conflicting evidence. Edward Teller was said to be taking bets on whether they would incinerate the whole atmosphere, instead of just a piece of it at Alamogordo NM with the first A-test.
Uh, Danman, I’m a cancer surgeon AND a cancer researcher. I’ve forgotten more about apoptosis to make room for new science and new findings on apoptosis than you know now or will ever know. Back in the late 1990s, although I didn’t work on a ceramide-related project, others in the lab did; so I heard about it during lab meetings every week.
Also, when it comes to biomarkers, it doesn’t matter what the biomarker is. It still has to be validated in clinical studies to demonstrate that it has prognostic significance or predicts response to therapy. I don’t care that it’s a “general marker for apoptosis.” That matters not one whit unless it’s clinically validated.
Real cancer researchers and cancer doctors understand that. You, obviously, do not.
Yes, but that’s not evidence that your particular theory is true. Don’t you see that I could use the same exact argument? “Well, there’s conflicting evidence like every scientific theory, but it’s definitely true without a doubt that there are no medicinal qualities to cannabis.”
Until there’s more research, we can’t make the kind of claims that you are. That’s why we need more research, particularly clinical trials. I believe that legalization, which should happen on its own merits, will lead to much higher quality research on this issue. That’s one of the reasons I support legalization.
What part of this do you have an issue with? Why is this unreasonable?
I live in the same state as AdamG, and I voted for legalization. I don’t care if you partake as long as you follow two simple requests:
1. Do it far away from me so I don’t have to smell the stench of its smoke.
2. Don’t make medical claims that you cannot support with verifiable citations (this assumes that you have actually read and understood the full paper).
Is that too much to ask?
By the way a couple of editorials from the local paper:
http://seattletimes.com/html/editorials/2024240972_editmarijuana05xml.html
http://blogs.seattletimes.com/opinionnw/2014/07/30/smoking-marijuana-in-front-of-children/
Read them, and please learn to not be a jerk.
Adam …….. within the first paragraph the author whips out the term “quackademic”.
You don`t see agenda there Adam?
You posted a review article that (1) you immediately demonstrated you couldn’t even understand, (2) devotes a whopping two paragraphs to the ceramide angle, and (3) isn’t really particularly interested in THC in the first place:
You just see “cannabinoid receptor” and stupidly jump to “weed!” Agonist, antagonist, you can’t be bothered to sort it out.
First of all, that’s in reference to an entirely different subject (But I bet you have some…interesting thoughts about TCM too). Second of all, In the very next paragraph Orac clearly states his position with regards to legalization, namely that he’s for it. So again, what is the agenda here? Why do you refuse to concretely state exactly what this supposed ‘agenda’ is?
Chris said; ….. “I live in the same state as AdamG, and I voted for legalization. I don’t care if you partake as long as you follow two simple requests:
1. Do it far away from me so I don’t have to smell the stench of its smoke.
2. Don’t make medical claims that you cannot support with verifiable citations (this assumes that you have actually read and understood the full paper).
A) You need to pay attention, understand, and acknowledge, that for the medicinal properties that I have cited, and many others I have not, which are supported by ABUNDANT PEER REVIEWED SCIENCE, along with countless thousands of patient reports worldwide, SMOKING WILL NOT BE EFFECTIVE …… do understand that? The plant needs to be EATEN …… NOT SMOKED.
B) I can sympathize with your right to not inhale second cannabis smoke, but PLEASE stop being a jerk and bellowing on about it.
within the first paragraph the author whips out the term “quackademic”.
You don`t see agenda there Adam?
“First of all, that’s in reference to an entirely different subject”
That`s called setting tone. Then, in the end he tries to link Cannabis Science to Chinese Herbalism, which he has already declared to be “quackademic” in the intro.
It`s a hit piece.
I’m sorry Danman, but you haven’t presented anything here that contradicts Orac’s claim that “it’s about belief first, and then trying to get the science to to support its magical properties.”
In fact, you’ve provided a perfect example. You presented a single article that, in it’s conclusion, states “Overall, the cannabinoids may show future promise in the treatment of cancer, but there are many significant hurdles to be overcome.”
All we’re doing is agreeing with the authors of your own citation. There are indeed many significant hurdles to overcome. You, however, have somehow twisted this article into definitive proof of medicinal benefit, without once acknowledging that more work is needed. In fact, you’ve basically condemned any further research. Isn’t this exactly the kind of thinking that Orac calls out?
Oh wow, this one’s in too deep. In addition to turning a list of “Peer-reviewed Articles on Cannabinoids and Cancer” into a list of 625 studies “in support” of the idea that “Cannabis Cures Cancer and whole lot of other diseases”
Danman even goes on to state
and
If that’s really how you feel about the current state of the literature you really have no business discussing this topic whatsoever. Please, I implore you, just give that last link a good read.
“You need to pay attention, understand, and acknowledge, that for the medicinal properties that I have cited, and many others I have not, which are supported by ABUNDANT PEER REVIEWED SCIENCE, along with countless thousands of patient reports worldwide, SMOKING WILL NOT BE EFFECTIVE …… do understand that? The plant needs to be EATEN …… NOT SMOKED. ”
Prove it. So far you have failed.
And you are being a jerk.
Peer Reviewed Cannabis and Cancer References (download PDF)
Argaw, A. et al. Concerted Action of CB1 Cannabinoid Receptor and Deleted in Colorectal Cancer in Axon Guidance. J Neurosci 31, 1489-1499 (2011).
Malfitano, A. M. et al. Update on the endocannabinoid system as an anticancer target. Expert Opin Ther Targets (2011).
Lorente, M. et al. Stimulation of the midkine/ALK axis renders glioma cells resistant to cannabinoid antitumoral action. Cell Death Differ (2011).
Karasu, T., Marczylo, T. H., Maccarrone, M. & Konje, J. C. The role of sex steroid hormones, cytokines and the endocannabinoid system in female fertility. Hum Reprod Update (2011).
Torres, S. et al. A Combined Preclinical Therapy of Cannabinoids and Temozolomide against Glioma. Mol Cancer Ther 10, 90-103 (2011).
Hsu, E. S. A review of granisetron, 5-hydroxytryptamine3 receptor antagonists, and other antiemetics. Am J Ther 17, 476-486 (2010).
Van Ryckeghem, F. & Van Belle, S. Management of chemotherapy-induced nausea and vomiting. Acta Clin Belg 65, 305-310 (2010).
Jordan, K. et al. Antiemetics in children receiving chemotherapy. MASCC/ESMO guideline update 2009. Support Care Cancer (2010).
Liu, W. M., Fowler, D. W. & Dalgleish, A. G. Cannabis-Derived Substances in Cancer Therapy – an Emerging Anti-Inflammatory Role for the Cannabinoids. Curr Clin Pharmacol (2010).
Larrinaga, G. et al. Cannabinoid CB1 Receptor Is Down-regulated in Clear Cell Renal Cell Carcinoma. J Histochem Cytochem (2010).
Lam, P. M., Marczylo, T. H. & Konje, J. C. Simultaneous measurement of three N- acylethanolamides in human bio-matrices using ultra performance liquid chromatography-tandem mass spectrometry. Anal Bioanal Chem (2010).
Tazi, E. & Errihani, H. Treatment of cachexia in oncology. Indian J Palliat Care 16, 129-137 (2010).
Thapa, D. et al. Novel hexahydrocannabinol analogs as potential anti-cancer agents inhibit cell proliferation and tumor angiogenesis. Eur J Pharmacol (2010).
Amoako, A. A. et al. Quantitative analysis of anandamide and related acylethanolamides in human seminal plasma by ultra performance liquid chromatography tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci (2010).
Peat, S. Using cannabinoids in pain and palliative care. Int J Palliat Nurs 16, 481- 485 (2010).
Hu, G., Ren, G. & Shi, Y. The putative cannabinoid receptor GPR55 promotes cancer cell proliferation. Oncogene (2010).
Preet, A. et al. Cannabinoid Receptors, CB1 and CB2, as Novel Targets for Inhibition of Non-Small Cell Lung Cancer Growth and Metastasis. Cancer Prev Res (Phila) (2010).
Saghafi, N., Lam, D. K. & Schmidt, B. L. Cannabinoids Attenuate Cancer Pain and Proliferation in a Mouse Model. Neurosci Lett (2010).
Karschner, E. L., Darwin, W. D., Goodwin, R. S., Wright, S. & Huestis, M. A. Plasma Cannabinoid Pharmacokinetics following Controlled Oral {Delta}9- Tetrahydrocannabinol and Oromucosal Cannabis Extract Administration. Clin Chem (2010).
Lin, H. C. et al. The relationship between endotoxemia and hepatic endocannabinoids in cirrhotic rats with portal hypertension. J Hepatol (2010).
Duran, M. et al. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br J Clin Pharmacol 70, 656-663 (2010).
Lee, T. Y., Lee, K. C. & Chang, H. H. Modulation of the cannabinoid receptors by andrographolide attenuates hepatic apoptosis following bile duct ligation in rats with fibrosis. Apoptosis (2010).
Yang, Q., Liu, H. Y., Zhang, Y. W., Wu, W. J. & Tang, W. X. Anandamide induces cell death through lipid rafts in hepatic stellate cells. J Gastroenterol Hepatol (2010).
Labar, G., Wouters, J. & Lambert, D. M. A Review on the Monoacylglycerol Lipase: At the Interface between Fat and Endocannabinoid Signalling. Curr Med Chem (2010).
Frampton, G., Coufal, M., Li, H., Ramirez, J. & Demorrow, S. Opposing actions of endocannabinoids on cholangiocarcinoma growth is via the differential activation of Notch signaling. Exp Cell Res (2010).
Xian, X. S. et al. Effect of a synthetic cannabinoid agonist on the proliferation and invasion of gastric cancer cells. J Cell Biochem (2010).
Jesus, M. L. et al. Opposite changes in cannabinoid CB(1) and CB(2) receptor expression in human gliomas. Neurochem Int (2010).
Laezza, C. et al. Inhibition of HMG-CoA reductase activity and of Ras farnesylation mediate antitumor effects of anandamide in human breast cancer cell. Endocr Relat Cancer (2010).
Wu, W. J. et al. [Membrane cholesterol mediates the endocannabinoids-anandamide effection on HepG2 cells.]. Zhonghua Gan Zang Bing Za Zhi 18, 204-208 (2010).
Fonseca, B. M. et al. N-Acylethanolamine Levels and Expression of Their Metabolizing Enzymes during Pregnancy. Endocrinology (2010).
Fernandez-Solari, J., Prestifilippo, J. P., Ossola, C. A., Rettori, V. & Elverdin, J. C. Participation of the endocannabinoid system in lipopolysaccharide-induced inhibition of salivary secretion. Arch Oral Biol (2010).
Whyte, D. A. et al. Cannabinoids Inhibit Cellular Respiration of Human Oral Cancer Cells. Pharmacology 85, 328-335 (2010).
Soneji, N. D., Paule, C. C., Mlynarczyk, M. & Nagy, I. Effects of cannabinoids on capsaicin receptor activity following exposure of primary sensory neurons to inflammatory mediators. Life Sci (2010).
Reiss, C. S. Cannabinoids and Viral Infections. Pharmaceuticals (Basel) 3, 1873- 1886 (2010).
Santha, P., Jenes, A., Somogyi, C. & Nagy, I. The endogenous cannabinoid anandamide inhibits transient receptor potential vanilloid type 1 receptor-mediated currents in rat cultured primary sensory neurons. Acta Physiol Hung 97, 149-158 (2010).
Ford, L. A. et al. A role for L-alpha-lysophosphatidylinositol and GPR55 in the modulation of migration, orientation and polarization of human breast cancer cells. Br J Pharmacol 160, 762-771 (2010).
Brown, I. et al. Cannabinoid receptor dependent and independent anti-proliferative effects of omega-3 ethanolamides in androgen receptor positive and negative prostate cancer cell lines. Carcinogenesis (2010).
Caffarel, M. M. et al. Cannabinoids reduce ErbB2-driven breast cancer progression through Akt inhibition. Mol Cancer 9, 196 (2010).
Park, J. Y. et al. Alcohol intake and risk of colorectal cancer: Results from the UK Dietary Cohort Consortium. Br J Cancer (2010).
Linsalata, M. et al. Effects of anandamide on polyamine levels and cell growth in human colon cancer cells. Anticancer Res 30, 2583-2589 (2010).
Marcu, J. P. et al. Cannabidiol Enhances the Inhibitory Effects of {Delta}9- Tetrahydrocannabinol on Human Glioblastoma Cell Proliferation and Survival. Mol Cancer Ther (2010).
Petrosino, S. & Di Marzo, V. FAAH and MAGL inhibitors: therapeutic opportunities from regulating endocannabinoid levels. Curr Opin Investig Drugs 11, 51-62 (2010).
Patel, K. D., Davison, J. S., Pittman, Q. J. & Sharkey, K. A. Cannabinoid CB(2) Receptors in Health and Disease. Curr Med Chem (2010).
Martini, L., Thompson, D., Kharazia, V. & Whistler, J. L. Differential Regulation of Behavioral Tolerance to WIN55,212-2 by GASP1. Neuropsychopharmacology (2010).
Gao, M., Wang, M., Miller, K. D., Hutchins, G. D. & Zheng, Q. H. Synthesis and in vitro biological evaluation of carbon-11-labeled quinoline derivatives as new candidate PET radioligands for cannabinoid CB2 receptor imaging. Bioorg Med Chem (2010).
Zoppi, S. et al. Regulatory Role of Cannabinoid Receptor 1 in Stress-Induced Excitotoxicity and Neuroinflammation. Neuropsychopharmacology (2010).
Cui, J. H. et al. Antinociceptive effect of intrathecal cannabinoid receptor agonist WIN 55,212-2 in a rat bone tumor pain model. Neurosci Lett (2010).
Kalifa, S., Polston, E. K., Allard, J. S. & Manaye, K. F. Distribution Patterns of Cannabinoid CB1 receptors in the hippocampus of APP(swe)/PS1(DeltaE9) double transgenic mice. Brain Res (2010).
De Petrocellis, L. et al. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br J Pharmacol (2010).
Miro, A. M. et al. 17beta-Estradiol regulates oxidative stress in prostate cancer cell lines according to ERalpha/ERbeta ratio. J Steroid Biochem Mol Biol (2010).
Nithipatikom, K., Isbell, M. A., Endsley, M. P., Woodliff, J. E. & Campbell, W. B. Anti-proliferative Effect of a Putative Endocannabinoid; 2-Arachidonylglyceryl Ether in Prostate Carcinoma Cells. Prostaglandins Other Lipid Mediat (2010).
Nunez, M., Perdomo, S., Moreta, J., Santos-Briz, A. & Gonzalez-Sarmiento, R. The G1359A-CNR1 gene polymorphism is associated to glioma in Spanish patients. Clin Transl Oncol 12, 825-828 (2010).
Ward, H., Luben, R. N., Wareham, N. J. & Khaw, K. T. CHD risk in relation to alcohol intake from categorical and open-ended dietary instruments. Public Health Nutr 1-8 (2010).
Yang, Y. Y., Liu, H., Nam, S. W., Kunos, G. & Lee, S. S. Mechanisms of TNFalpha-induced cardiac dysfunction in cholestatic bile duct-ligated mice: interaction between TNFalpha and endocannabinoids. J Hepatol 53, 298-306 (2010).
Miranville, A., Herling, A. W., Biemer-Daub, G. & Voss, M. D. Reversal of Inflammation-Induced Impairment of Glucose Uptake in Adipocytes by Direct Effect of CB1 Antagonism on Adipose Tissue Macrophages. Obesity (Silver Spring) (2010).
Kawamata, T., Niiyama, Y., Yamamoto, J. & Furuse, S. Reduction of bone cancer pain by CB1 activation and TRPV1 inhibition. J Anesth (2010).
Fowler, C. J. et al. Targeting the Endocannabinoid System for the Treatment of Cancer – A Practical View. Curr Top Med Chem (2010).
Taylor, A. H., Abbas, M. S., Habiba, M. A. & Konje, J. C. Histomorphometric evaluation of cannabinoid receptor and anandamide modulating enzyme expression in the human endometrium through the menstrual cycle. Histochem Cell Biol (2010).
Gallotta, D. et al. Rimonabant-induced apoptosis in leukemia cell lines: activation of caspase-dependent and -independent pathways. Biochem Pharmacol (2010).
Mukhopadhyay, B. et al. Transcriptional regulation of cannabinoid receptor-1 expression in the liver by retinoic acid acting via retinoic acid receptor-{gamma}. J Biol Chem (2010).
De Laurentiis, A., Fernandez Solari, J., Mohn, C., Zorrilla Zubilete, M. & Rettori, V. Endocannabinoid System Participates in Neuroendocrine Control of Homeostasis. Neuroimmunomodulation 17, 153-156 (2010).
Thors, L. et al. Fatty Acid Amide Hydrolase in Prostate Cancer: Association with Disease Severity and Outcome, CB(1) Receptor Expression and Regulation by IL-4. PLoS One 5, (2010).
Phillips, R. S. et al. Antiemetic medication for prevention and treatment of chemotherapy induced nausea and vomiting in childhood. Cochrane Database Syst Rev 9, CD007786 (2010).
Olea-Herrero, N., Vara, D., Malagarie-Cazenave, S. & Diaz-Laviada, I. The cannabinoid R(+)methanandamide induces IL-6 secretion by prostate cancer PC3 cells. J Immunotoxicol (2009).
Izzo, A. A., Borrelli, F., Capasso, R., Di Marzo, V. & Mechoulam, R. Non- psychotropic plant cannabinoids: new therapeutic opportunities from an ancient herb. Trends Pharmacol Sci (2009).
Wainwright, C. & McGrath, J. Editorial. Br J Pharmacol 158, 393-394 (2009).
Guindon, J. & Hohmann, A. G. The Endocannabinoid System and Pain. CNS
Neurol Disord Drug Targets (2009).
Horn, C. C., Still, L., Fitzgerald, C. & Friedman, M. I. Food restriction, refeeding, and gastric fill fail to affect emesis in musk shrews. Am J Physiol Gastrointest Liver Physiol (2009).
Freimuth, N., Ramer, R. & Hinz, B. Antitumorigenic effects of cannabinoids beyond apoptosis. J Pharmacol Exp Ther (2009).
Tournier, N. et al. Interaction of drugs of abuse and maintenance treatments with human P-glycoprotein (ABCB1) and breast cancer resistance protein (ABCG2). Int J Neuropsychopharmacol 1-11 (2009).
Johnson, J. R. et al. Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study of the Efficacy, Safety, and Tolerability of THC:CBD Extract and THC Extract in Patients With Intractable Cancer-Related Pain. J Pain Symptom Manage (2009).
Qamri, Z. et al. Synthetic cannabinoid receptor agonists inhibit tumor growth and metastasis of breast cancer. Mol Cancer Ther (2009).
Rask-Andersen, M., Olszewski, P. K., Levine, A. S. & Schioth, H. B. Molecular mechanisms underlying anorexia nervosa: Focus on human gene association studies and systems controlling food intake. Brain Res Rev (2009).
De Filippis, D. et al. LEVELS OF ENDOCANNABINOIDs AND PALMITOYLETHANOLAMIDE AND THEIR PHARMACOLOGICAL MANIPULATION IN CHRONIC GRANULOMATOUS INFLAMMATION IN RATs. Pharmacol Res (2009).
Leelawat, S., Leelawat, K., Narong, S. & Matangkasombut, O. The Dual Effects of Delta(9)-Tetrahydrocannabinol on Cholangiocarcinoma Cells: Anti-Invasion Activity at Low Concentration and Apoptosis Induction at High Concentration. Cancer Invest (2009).
Ramer, R., Merkord, J., Rohde, H. & Hinz, B. Cannabidiol Inhibits Cancer Cell Invasion Via Upregulation Of Tissue Inhibitor Of Matrix Metalloproteinases-1. Biochem Pharmacol (2009).
Brighton, P. J. et al. Characterization of anandamide-stimulated cannabinoid receptor signaling in human ULTR myometrial smooth muscle cells. Mol Endocrinol (2009).
Kingsley, P. J. & Marnett, L. J. Analysis of endocannabinoids, their congeners and COX-2 metabolites. J Chromatogr B Analyt Technol Biomed Life Sci (2009).
Takeda, S., Yamamoto, I. & Watanabe, K. Modulation of Delta(9)- tetrahydrocannabinol-induced MCF-7 breast cancer cell growth by cyclooxygenase and aromatase. Toxicology 259, 25-32 (2009).
Cotter, J. Efficacy of Crude Marijuana and Synthetic Delta-9-Tetrahydrocannabinol as Treatment for Chemotherapy-Induced Nausea and Vomiting: A Systematic Literature Review. Oncol Nurs Forum 36, 345-352 (2009).
Wegener, N. & Koch, M. Neurobiology and Systems Physiology of the Endocannabinoid System. Pharmacopsychiatry 42, S79-S86 (2009)
Brizzi, A. et al. New Resorcinol-Anandamide “Hybrids” as Potent Cannabinoid Receptor Ligands Endowed with Antinociceptive Activity in Vivo. J Med Chem (2009).
Izzo, A. A. & Camilleri, M. Cannabinoids in intestinal inflammation and cancer. Pharmacol Res (2009).
Yin, H. et al. Lipid G-protein-coupled Receptor Ligand Identification Using beta – arrestin Pathhunter Assay. J Biol Chem (2009).
Villanueva, A. et al. Central Cannabinoid-1 Receptor Antagonist Administration Prevents Endotoxic Hypotension Affecting Norepinephrine Release in the Preoptic Anterior Hypothalamic Area. Shock (2009).
Farquhar-Smith, W. P. Do cannabinoids have a role in cancer pain management? Curr Opin Support Palliat Care 3, 7-13 (2009).
Navari, R. M. Antiemetic control: toward a new standard of care for emetogenic chemotherapy. Expert Opin Pharmacother 10, 629-644 (2009).
Oesch, S. et al. Cannabinoid receptor 1 is a potential drug target for treatment of translocation-positive rhabdomyosarcoma. Mol Cancer Ther (2009).
Furuse, S. et al. Reduction of Bone Cancer Pain by Activation of Spinal Cannabinoid Receptor 1 and Its Expression in the Superficial Dorsal Horn of the Spinal Cord in a Murine Model of Bone Cancer Pain. Anesthesiology (2009).
Schley, M. et al. Predominant CB2 receptor expression in endothelial cells of glioblastoma in humans. Brain Res Bull 79, 333-337 (2009).
Udler, M. S. et al. Common germline polymorphisms in COMT, CYP19A1, ESR1, PGR, SULT1E1 and STS and survival after a diagnosis of breast cancer. Int J Cancer (2009).
Luca, T. et al. The CB(1)/CB(2) receptor agonist WIN-55,212-2 reduces viability of human Kaposi’s sarcoma cells in vitro. Eur J Pharmacol (2009).
Galal, A. M. et al. Naturally Occurring and Related Synthetic Cannabinoids and their Potential Therapeutic Applications. Recent Pat CNS Drug Discov 4, 112-136 (2009).
Liang, C. et al. A Population-Based Case-Control Study of Marijuana Use and Head and Neck Squamous Cell Carcinoma. Cancer Prev Res (Phila Pa) (2009).
Surtees, P. G., Wainwright, N. W., Luben, R. N., Khaw, K. T. & Bingham, S. A. No evidence that social stress is associated with breast cancer incidence. Breast Cancer Res Treat (2009).
Biro, T., Toth, B. I., Hasko, G., Paus, R. & Pacher, P. The endocannabinoid system of the skin in health and disease: novel perspectives and therapeutic opportunities. Trends Pharmacol Sci (2009).
Han, K. H. et al. CB1 and CB2 Cannabinoid Receptors Differentially Regulate the Production of Reactive Oxygen Species by Macrophages. Cardiovasc Res (2009).
Oesch, S. & Gertsch, J. Cannabinoid receptor ligands as potential anticancer agents – high hopes for new therapies? J Pharm Pharmacol 61, 839-853 (2009).
Gustafsson, K., Sander, B., Bielawski, J., Hannun, Y. A. & Flygare, J. Potentiation of cannabinoid-induced cytotoxicity in mantle cell lymphoma through modulation of ceramide metabolism. Mol Cancer Res 7, 1086-1098 (2009).
Ramesh, Shukla, N. K. & Bhatnagar, S. Phantom breast syndrome. Indian J Palliat Care 15, 103-107 (2009).
Surtees, P. G. et al. No association between APOE and major depressive disorder in a community sample of 17,507 adults. J Psychiatr Res (2009).
Dunning, A. M. et al. Association of ESR1 gene tagging SNPs with breast cancer risk. Hum Mol Genet (2009).
Van Dross, R. T. Metabolism of anandamide by COX-2 is necessary for endocannabinoid-induced cell death in tumorigenic keratinocytes. Mol Carcinog (2009).
Lorente, M. et al. Amphiregulin is a factor for resistance of glioma cells to cannabinoid-induced apoptosis. Glia (2009).
Pisanti, S. et al. Use of cannabinoid receptor agonists in cancer therapy as palliative and curative agents. Best Pract Res Clin Endocrinol Metab 23, 117-131 (2009).
Froger, N. et al. Cannabinoids prevent the opposite regulation of astroglial connexin43 hemichannels and gap junction channels induced by pro-inflammatory treatments. J Neurochem 111, 1383-1397 (2009).
Madeddu, C. & Mantovani, G. An update on promising agents for the treatment of cancer cachexia. Curr Opin Support Palliat Care (2009).
Mantovani, G. & Madeddu, C. Cancer cachexia: medical management. Support Care Cancer (2009).
Olea-Herrero, N., Vara, D., Malagarie-Cazenave, S. & Diaz-Laviada, I. Inhibition of human tumour prostate PC-3 cell growth by cannabinoids R(+)-Methanandamide and JWH-015: Involvement of CB(2). Br J Cancer (2009).
Zhao, P., Leonoudakis, D., Abood, M. E. & Beattie, E. Cannabinoid Receptor Activation Reduces TNFalpha-Induced Surface Localization of AMPAR-Type Glutamate Receptors and Excitotoxicity. Neuropharmacology (2009).
Pisanti, S. & Bifulco, M. Endocannabinoid system modulation in cancer biology and therapy. Pharmacol Res 60, 107-116 (2009).
Moayyeri, A. et al. Estimation of absolute fracture risk among middle-aged and older men and women: the EPIC-Norfolk population cohort study. Eur J Epidemiol (2009).
Santoro, A. et al. Rimonabant inhibits human colon cancer cell growth and reduces the formation of precancerous lesions in the mouse colon. Int J Cancer (2009).
Wang, Q. et al. Pretreatment With Electroacupuncture Induces Rapid Tolerance to Focal Cerebral Ischemia Through Regulation of Endocannabinoid System. Stroke (2009).
Gallant, M., Odei-Addo, F., Frost, C. L. & Levendal, R. A. Biological effects of THC and a lipophilic cannabis extract on normal and insulin resistant 3T3-L1 adipocytes. Phytomedicine (2009).
Salazar, M. et al. Cannabinoid action induces autophagy-mediated cell death through stimulation of ER stress in human glioma cells. J Clin Invest (2009).
El-Talatini, M. R. et al. Localisation and function of the endocannabinoid system in the human ovary. PLoS ONE 4, e4579 (2009).
Keeley, P. W. Nausea and vomiting in people with cancer and other chronic diseases. Clin Evid (Online) 2009, (2009).
Alpini, G. & Demorrow, S. Chapter 18 Changes in the Endocannabinoid System May Give Insight into new and Effective Treatments for Cancer. Vitam Horm 81C, 469-485 (2009).
Bifulco, M., Santoro, A., Laezza, C. & Malfitano, A. M. Chapter 7 Cannabinoid Receptor CB1 Antagonists State of the Art and Challenges. Vitam Horm 81, 159- 189 (2009).
Wang, J. et al. Expression and secretion of N-acylethanolamine-hydrolyzing acid amidase in human prostate cancer cells. J Biochem (2008).
Zuardi, A. W. Cannabidiol: from an inactive cannabinoid to a drug with wide spectrum of action. Rev Bras Psiquiatr 30, 271-280 (2008).
Khasabova, I. A. et al. A decrease in anandamide signaling contributes to the maintenance of cutaneous mechanical hyperalgesia in a model of bone cancer pain. J Neurosci 28, 11141-11152 (2008).
Eichele, K., Ramer, R. & Hinz, B. R(+)-Methanandamide-Induced Apoptosis of Human Cervical Carcinoma Cells Involves A Cyclooxygenase-2-Dependent Pathway. Pharm Res (2008).
Demorrow, S. et al. Anandamide inhibits cholangiocyte hyperplastic proliferation via activation of thioredoxin 1/redox factor 1 and AP-1 activation. Am J Physiol Gastrointest Liver Physiol 294, G506-19 (2008).
Van der Kooy, F., Pomahacova, B. & Verpoorte, R. Cannabis Smoke Condensate II: Influence of Tobacco on Tetrahydrocannabinol Levels. Inhal Toxicol 1 (2008).
Burstein, S. & Salmonsen, R. Acylamido analogs of endocannabinoids selectively inhibit cancer cell proliferation. Bioorg Med Chem (2008).
Czifra, G. et al. Increased expressions of cannabinoid receptor-1 and transient receptor potential vanilloid-1 in human prostate carcinoma. J Cancer Res Clin Oncol (2008).
Lissoni, P. et al. The endocannabinoid anandamide neither impairs in vitro T-cell function nor induces regulatory T-cell generation. Anticancer Res 28, 3743-3748 (2008).
Giuliano, M. et al. Apoptosis induced in HepG2 cells by the synthetic cannabinoid WIN: Involvement of the transcription factor PPARgamma. Biochimie (2008).
Purohit, V., Gao, B. & Song, B. J. Molecular Mechanisms of Alcoholic Fatty Liver. Alcohol Clin Exp Res (2008).
Caffarel, M. M. et al. JunD is involved in the antiproliferative effect of Delta(9)- tetrahydrocannabinol on human breast cancer cells. Oncogene (2008).
Casida, J. E., Nomura, D. K., Vose, S. C. & Fujioka, K. Organophosphate-sensitive lipases modulate brain lysophospholipids, ether lipids and endocannabinoids. Chem Biol Interact (2008).
Buznikov, G. A. et al. Amyloid Precursor Protein 96-110 and Beta-amyloid 1-42 Elicit Developmental Anomalies in Sea Urchin Embryos and Larvae That Are Alleviated By Neurotransmitter Analogs for Acetylcholine, Serotonin and Cannabinoids. Neurotoxicol Teratol (2008).
Zheng, D. et al. The cannabinoid receptors are required for ultraviolet-induced inflammation and skin cancer development. Cancer Res 68, 3992-3998 (2008).
Deutsch, S. I. et al. Current Status of Cannabis Treatment of Multiple Sclerosis with an Illustrative Case Presentation of a Patient with MS, Complex Vocal Tics, Paroxysmal Dystonia, and Marijuana Dependence Treated with Dronabinol. CNS Spectr 13, 393-403 (2008).
Surtees, P. G. et al. Psychological distress, major depressive disorder, and risk of stroke. Neurology 70, 788-794 (2008).
De Petrocellis, L. et al. Plant-derived cannabinoids modulate the activity of transient receptor potential channels of ankyrin type-1 (TRPA1) and melastatin type-8 (TRPM-8). J Pharmacol Exp Ther (2008).
Blazquez, C. et al. Cannabinoids inhibit glioma cell invasion by down-regulating matrix metalloproteinase-2 expression. Cancer Res 68, 1945-1952 (2008).
Endsley, M. P. et al. Expression and Function of Fatty Acid Amide Hydrolase in Prostate Cancer. Int J Cancer (2008).
Iribarne, M. et al. Cannabinoid Receptors in Conjunctival Epithelium: Identification and Functional Properties. Invest Ophthalmol Vis Sci (2008).
Gustafsson, K. et al. Expression of Cannabinoid Receptors Type 1 and Type 2 in Non-hodkin Lymphoma: Growth Inhibition By Receptor Activation. Int J Cancer (2008).
Bifulco, M., Malfitano, A. M., Pisanti, S. & Laezza, C. Endocannabinoids in endocrine and related tumours. Endocr Relat Cancer 15, 391-408 (2008).
Widmer, M., Hanemann, O. & Zajicek, J. High concentrations of cannabinoids activate apoptosis in human U373MG glioma cells. J Neurosci Res (2008).
Habayeb, O. M., Taylor, A. H., Bell, S. C., Taylor, D. J. & Konje, J. C. Expression of the Endocannabinoid System in Human First Trimester Placenta and its Role in Trophoblast Proliferation. Endocrinology (2008).
Machado Rocha, F. C., Stefano, S. C., DE Cassia Haiek, R., Rosa Oliveira, L. M. & DA Silveira, D. X. Therapeutic use of Cannabis sativa on chemotherapy-induced nausea and vomiting among cancer patients: systematic review and meta-analysis. Eur J Cancer Care (Engl) (2008).
Rahn, E. J. et al. Selective Activation of Cannabinoid CB2 Receptors Suppresses Neuropathic Nociception Induced by Treatment with the Chemotherapeutic Agent Paclitaxel in Rats. J Pharmacol Exp Ther (2008).
van Diepen, H., Schlicker, E. & Michel, M. C. Prejunctional and peripheral effects of the cannabinoid CB(1) receptor inverse agonist rimonabant (SR 141716). Naunyn Schmiedebergs Arch Pharmacol (2008).
Gustafsson, S. B., Lindgren, T., Jonsson, M. & Jacobsson, S. O. Cannabinoid receptor-independent cytotoxic effects of cannabinoids in human colorectal carcinoma cells: synergism with 5-fluorouracil. Cancer Chemother Pharmacol (2008).
von Bueren, A. O., Schlumpf, M. & Lichtensteiger, W. Delta(9)- tetrahydrocannabinol inhibits 17beta-estradiol-induced proliferation and fails to activate androgen and estrogen receptors in MCF7 human breast cancer cells. Anticancer Res 28, 85-89 (2008).
Ramer, R. & Hinz, B. Inhibition of cancer cell invasion by cannabinoids via increased expression of tissue inhibitor of matrix metalloproteinases-1. J Natl Cancer Inst 100, 59-69 (2008).
Sarfaraz, S., Adhami, V. M., Syed, D. N., Afaq, F. & Mukhtar, H. Cannabinoids for cancer treatment: progress and promise. Cancer Res 68, 339-342 (2008).
Warr, D. G. Chemotherapy- and cancer-related nausea and vomiting. Curr Oncol 15, S4-9 (2008).
Jones, J. D., Carney, S. T., Vrana, K. E., Norford, D. C. & Howlett, A. C. Cannabinoid receptor-mediated translocation of NO-sensitive guanylyl cyclase and production of cyclic GMP in neuronal cells. Neuropharmacology 54, 23-30 (2008).
Bari, M. et al. Type-1 cannabinoid receptors colocalize with caveolin-1 in neuronal cells. Neuropharmacology 54, 45-50 (2008).
Blazquez, C. et al. Down-regulation of tissue inhibitor of metalloproteinases-1 in gliomas: a new marker of cannabinoid antitumoral activity? Neuropharmacology 54, 235-243 (2008).
Notarnicola, M. et al. Estrogenic induction of cannabinoid CB1 receptor in human colon cancer cell lines. Scand J Gastroenterol 43, 66-72 (2008).
Shi, Y. et al. Cannabinoid 2 receptor induction by IL-12 and its potential as a therapeutic target for the treatment of anaplastic thyroid carcinoma. Cancer Gene Ther 15, 101-107 (2008).
Massi, P. et al. 5-Lipoxygenase and anandamide hydrolase (FAAH) mediate the antitumor activity of cannabidiol, a non-psychoactive cannabinoid. J Neurochem 104, 1091-1100 (2008).
Rudolph, M. I. et al. The Influence of Mast Cell Mediators on Migration of SW756 Cervical Carcinoma Cells. J Pharmacol Sci 106, 208-218 (2008).
Ware1, M. A., Daeninck, P. & Maida, V. A review of nabilone in the treatment of chemotherapy-induced nausea and vomiting. Ther Clin Risk Manag 4, 99-107 (2008).
Russo, E. B. Cannabinoids in the management of difficult to treat pain. Ther Clin Risk Manag 4, 245-259 (2008).
Diaz, P. et al. 6-Methoxy-N-alkyl Isatin Acylhydrazone Derivatives as a Novel Series of Potent Selective Cannabinoid Receptor 2 Inverse Agonists: Design, Synthesis, and Binding Mode Prediction. J Med Chem (2008).
Sugamura, K. et al. Activated Endocannabinoid System in Coronary Artery Disease and Antiinflammatory Effects of Cannabinoid 1 Receptor Blockade on Macrophages. Circulation (2008).
Chung, S. C. et al. A high cannabinoid CB(1) receptor immunoreactivity is associated with disease severity and outcome in prostate cancer. Eur J Cancer (2008).
Cianchi, F. et al. Cannabinoid Receptor Activation Induces Apoptosis through Tumor Necrosis Factor {alpha}-Mediated Ceramide De novo Synthesis in Colon Cancer Cells. Clin Cancer Res 14, 7691-7700 (2008).
Anand, P., Whiteside, G., Fowler, C. J. & Hohmann, A. G. Targeting CB(2) receptors and the endocannabinoid system for the treatment of pain. Brain Res Rev (2008).
Yazulla, S. Endocannabinoids in the retina: From marijuana to neuroprotection. Prog Retin Eye Res (2008).
Wang, D. et al. Loss of cannabinoid receptor 1 accelerates intestinal tumor growth. Cancer Res 68, 6468-6476 (2008).
Santoro, A. et al. Reply to the letter to the editor “Long-term cannabinoid receptor (CB1) blockade in obesity: Implications for the development of colorectal cancer”. Int J Cancer (2008).
Potenzieri, C., Harding-Rose, C. & Simone, D. A. The cannabinoid receptor agonist, WIN 55, 212-2, attenuates tumor-evoked hyperalgesia through peripheral mechanisms. Brain Res (2008).
Ernst, G. & Kongsgaard, U. E. [Use of cannabinoids in palliative medicine.]. Tidsskr Nor Laegeforen 128, 822-825 (2008).
DUBOIS. Turned-off Cannabinoid Receptor Turns on Colorectal Tumor Growth. Newswise (2008).
Bilsland, L. G. & Greensmith, L. The endocannabinoid system in amyotrophic lateral sclerosis. Curr Pharm Des 14, 2306-2316 (2008).
Massi, P., Valenti, M., Bolognini, D. & Parolaro, D. Expression and function of the endocannabinoid system in glial cells. Curr Pharm Des 14, 2289-2298 (2008).
Galanti, G. et al. Delta 9-tetrahydrocannabinol inhibits cell cycle progression by downregulation of E2F1 in human glioblastoma multiforme cells. Acta Oncol 47, 1062-1070 (2008).
Izzo, A. A. et al. Increased endocannabinoid levels reduce the development of precancerous lesions in the mouse colon. J Mol Med (2007).
Izzo, A. A. The cannabinoid CB(2) receptor: a good friend in the gut. Neurogastroenterol Motil 19, 704-708 (2007).
Peters, M. & Kogan, N. M. HU-331: a cannabinoid quinone, with uncommon cytotoxic properties and low toxicity. Expert Opin Investig Drugs 16, 1405-1413 (2007).
Michalski, C. W. et al. Cannabinoids in pancreatic cancer: Correlation with survival and pain. Int J Cancer (2007).
Holland, M. L., Lau, D. T., Allen, J. D. & Arnold, J. C. The multidrug transporter ABCG2 (BCRP) is inhibited by plant-derived cannabinoids. Br J Pharmacol (2007).
Mukhopadhyay, S. & Tulis, D. A. Endocannabinoid regulation of matrix metalloproteinases: implications in ischemic stroke. Cardiovasc Hematol Agents Med Chem 5, 311-318 (2007).
Gary-Bobo, M. et al. Rimonabant reduces obesity-associated hepatic steatosis and features of metabolic syndrome in obese Zucker fa/fa rats. Hepatology (2007).
Seamon, M. J., Fass, J. A., Maniscalco-Feichtl, M. & Abu-Shraie, N. A. Medical marijuana and the developing role of the pharmacist. Am J Health Syst Pharm 64, 1037-1044 (2007).
Varvel, S. A., Martin, B. R. & Lichtman, A. H. Lack of behavioral sensitization after repeated exposure to THC in mice and comparison to methamphetamine. Psychopharmacology (Berl) (2007).
Slatkin, N. E. Cannabinoids in the treatment of chemotherapy-induced nausea and vomiting: beyond prevention of acute emesis. J Support Oncol 5, 1-9 (2007).
Tham, C. S., Whitaker, J., Luo, L. & Webb, M. Inhibition of microglial fatty acid amide hydrolase modulates LPS stimulated release of inflammatory mediators. FEBS Lett 581, 2899-2904 (2007).
Taylor, A. H., Ang, C., Bell, S. C. & Konje, J. C. The role of the endocannabinoid system in gametogenesis, implantation and early pregnancy. Hum Reprod Update (2007).
Greenhough, A., Patsos, H. A., Williams, A. C. & Paraskeva, C. The cannabinoid delta(9)-tetrahydrocannabinol inhibits RAS-MAPK and PI3K-AKT survival signalling and induces BAD-mediated apoptosis in colorectal cancer cells. Int J Cancer (2007).
Gkoumassi, E. et al. Virodhamine and CP55,940 modulate cAMP production and IL-8 release in human bronchial epithelial cells. Br J Pharmacol (2007).
Preet, A., Ganju, R. K. & Groopman, J. E. Delta(9)-Tetrahydrocannabinol inhibits epithelial growth factor-induced lung cancer cell migration in vitro as well as its growth and metastasis in vivo. Oncogene (2007).
Vila, A., Rosengarth, A., Piomelli, D., Cravatt, B. & Marnett, L. J. Hydrolysis of Prostaglandin Glycerol Esters by the Endocannabinoid-Hydrolyzing Enzymes, Monoacylglycerol Lipase and Fatty Acid Amide Hydrolase. Biochemistry (2007).
Udler, M. et al. Common germline genetic variation in antioxidant defense genes and survival after diagnosis of breast cancer. J Clin Oncol 25, 3015-3023 (2007).
Schramm-Sapyta, N. L. et al. Differential anxiogenic, aversive, and locomotor effects of THC in adolescent and adult rats. Psychopharmacology (Berl) S (2007).
Jordan, K., Schmoll, H. J. & Aapro, M. S. Comparative activity of antiemetic drugs. Crit Rev Oncol Hematol S (2007).
Aguado, T. et al. Cannabinoids induce glioma stem-like cell differentiation and inhibit gliomagenesis. J Biol Chem S (2007).
Demorrow, S. et al. Opposing actions of endocannabinoids on cholangiocarcinoma growth: Recruitment of fas and fas ligand to lipid rafts. J Biol Chem S (2007).
Bifulco, M., Grimaldi, C., Gazzerro, P., Pisanti, S. & Santoro, A. Rimonabant: Just an Anti-obesity Drug? Current Evidence on Its Pleiotropic Effects. Mol Pharmacol (2007).
Darmani, N. A., Janoyan, J. J., Crim, J. & Ramirez, J. Receptor mechanism and antiemetic activity of structurally-diverse cannabinoids against radiation-induced emesis in the least shrew. Eur J Pharmacol S (2007).
Fogarty, A. et al. Marijuana as therapy for people living with HIV/AIDS: Social and health aspects. AIDS Care S 19, 295-301 (2007).
Athanasiou, A. et al. Cannabinoid receptor agonists are mitochondrial inhibitors: A unified hypothesis of how cannabinoids modulate mitochondrial function and induce cell death. Biochem Biophys Res Commun 364, 131-137 (2007).
Takeda, S. et al. Delta(9)-Tetrahydrocannabinol enhances MCF-7 cell proliferation via cannabinoid receptor-independent signaling. Toxicology (2007).
Flygare, J. & Sander, B. The endocannabinoid system in cancer-Potential therapeutic target? Semin Cancer Biol (2007).
Engels, F. K. et al. Medicinal cannabis in oncology. Eur J Cancer 43, 2638-2644 (2007).
Russo, E. B., Guy, G. W. & Robson, P. J. Cannabis, Pain, and Sleep: Lessons from Therapeutic Clinical Trials of Sativex((R)), a Cannabis-Based Medicine. Chem Biodivers 4, 1729-1743 (2007).
Zhang, X., Wang, J. F., Kunos, G. & Groopman, J. E. Cannabinoid Modulation of Kaposi’s Sarcoma Associated Herpesvirus Infection and Transformation. Cancer Res 67, 7230-7237 (2007).
Kempf, K. et al. Immune-mediated Activation of the Endocannabinoid System in Visceral Adipose Tissue in Obesity. Horm Metab Res 39, 596-600 (2007).
Batkai, S. et al. Decreased age-related cardiac dysfunction, myocardial nitrative stress, inflammatory gene expression, and apoptosis in mice lacking fatty acid amide hydrolase. Am J Physiol Heart Circ Physiol 293, H909-18 (2007).
Velasco, G. et al. Cannabinoids and gliomas. Mol Neurobiol 36, 60-67 (2007).
De Filippis, D. et al. Local administration of WIN 55,212-2 reduces chronic granuloma-associated angiogenesis in rat by inhibiting NF-kappaB activation. J Mol Med (2007).
Bifulco, M., Laezza, C., Gazzerro, P. & Pentimalli, F. Endocannabinoids as emerging suppressors of angiogenesis and tumor invasion (Review). Oncol Rep 17, 813-816 (2007).
D’Souza, D. C. Cannabinoids and psychosis. Int Rev Neurobiol S 78, 289-326 (2007).
Whiteside, G. T., Lee, G. P. & Valenzano, K. J. The role of the cannabinoid CB2 receptor in pain transmission and therapeutic potential of small molecule CB2 receptor agonists. Curr Med Chem 14, 917-936 (2007).
Sunil K. Aggarwal, M. S. I. I. I., PhC, BS, BA, Muraco Kyashna-Tocha, PhD; Gregory T. Carter, MD, MS. Dosing Medical Marijuana: Rational Guidelines on Trial in Washington State. (2007).
Kingsley, P. J. & Marnett, L. J. LC-MS-MS Analysis of Neutral Eicosanoids. Methods Enzymol 433, 91-112 (2007).
Kogan, N. M. & Mechoulam, R. Cannabinoids in health and disease. Dialogues Clin Neurosci 9, 413-430 (2007).
Goanvic, D. L. & Tius, M. A. Oxaza adamantyl cannabinoids. A new class of cannabinoid receptor probes. J Org Chem 71, 7800-7804 (2006).
Pacher, P., Batkai, S. & Kunos, G. The endocannabinoid system as an emerging target of pharmacotherapy. Pharmacol Rev 58, 389-462 (2006).
Karanian, D. A. & Bahr, B. A. Cannabinoid drugs and enhancement of endocannabinoid responses: strategies for a wide array of disease states. Curr Mol Med 6, 677-684 (2006).
Ziring, D. et al. Formation of B and T cell subsets require the cannabinoid receptor CB2. Immunogenetics 58, 714-725 (2006).
Ligresti, A. et al. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. J Pharmacol Exp Ther 318, 1375- 1387 (2006).
Sarfaraz, S., Afaq, F., Adhami, V. M., Malik, A. & Mukhtar, H. Cannabinoid receptor agonist induced apoptosis of human prostate cancer cells LNCaP proceeds through sustained activation of ERK1/2 leading to G1 cell cycle arrest. J Biol Chem (2006).
Blazquez, C. et al. Cannabinoid receptors as novel targets for the treatment of melanoma. FASEB J (2006).
Stokes, A. et al. TRPA1 is a substrate for de-ubiquitination by the tumor suppressor CYLD. Cell Signal 18, 1584-1594 (2006).
De Lago, E. et al. Acyl-based anandamide uptake inhibitors cause rapid toxicity to C6 glioma cells at pharmacologically relevant concentrations. J Neurochem 99, 677-688 (2006).
Xu, X. et al. Overexpression of cannabinoid receptors CB1 and CB2 correlates with improved prognosis of patients with hepatocellular carcinoma. Cancer Genet Cytogenet 171, 31-38 (2006).
Fernandez-Solari, J., Prestifilippo, J. P., Bornstein, S. R., McCann, S. M. & Rettori, V. Participation of the Endocannabinoid System in the Effect of TNF-{alpha} on Hypothalamic Release of Gonadotropin-Releasing Hormone. Ann N Y Acad Sci S 1088, 238-250 (2006).
Mormina, M. E., Thakur, S., Molleman, A., Whelan, C. J. & Baydoun, A. R. Cannabinoid signalling in TNF-alpha induced IL-8 release. Eur J Pharmacol (2006).
Raduner, S. et al. Alkylamides from Echinacea Are a New Class of Cannabinomimetics: CANNABINOID TYPE 2 RECEPTOR-DEPENDENT AND – INDEPENDENT IMMUNOMODULATORY EFFECTS. J Biol Chem 281, 14192- 14206 (2006).
Alsasua Del Valle, A. Implication of Cannabinoids in Neurological Diseases. Cell Mol Neurobiol (2006).
Bifulco, M., Laezza, C., Pisanti, S. & Gazzerro, P. Cannabinoids and cancer: pros and cons of an antitumour strategy. Br J Pharmacol 148, 123-135 (2006).
Kogan, N. M. et al. A Cannabinoid Quinone Inhibits Angiogenesis by Targeting Vascular Endothelial Cells. Mol Pharmacol (2006).
Fogli, S. et al. Cannabinoid derivatives induce cell death in pancreatic MIA PaCa-2 cells via a receptor-independent mechanism. FEBS Lett 580, 1733-1739 (2006).
Bari, M., Battista, N., Fezza, F., Gasperi, V. & Maccarrone, M. New insights into endocannabinoid degradation and its therapeutic potential. Mini Rev Med Chem 6, 257-268 (2006).
Fernandez-Lopez, D. et al. Characterization of the Neuroprotective Effect of the Cannabinoid Agonist WIN-55212 in an In Vitro Model of Hypoxic-Ischemic Brain Damage in Newborn Rats. Pediatr Res (2006).
Guzman, M. et al. A pilot clinical study of Delta(9)-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer (2006).
Sarnataro, D. et al. The Cannabinoid CB1 Receptor Antagonist Rimonabant (SR141716) Inhibits Human Breast Cancer Cell Proliferation Through A Lipid Rafts Mediated Mechanism. Mol Pharmacol (2006).
Strasser, F. et al. Comparison of orally administered cannabis extract and delta-9- tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: a multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J Clin Oncol 24, 3394- 3400 (2006).
Carracedo, A. et al. Cannabinoids Induce Apoptosis of Pancreatic Tumor Cells via Endoplasmic Reticulum Stress-Related Genes. Cancer Res 66, 6748-6755 (2006).
Caffarel, M. M., Sarrio, D., Palacios, J., Guzman, M. & Sanchez, C. Delta}9- Tetrahydrocannabinol Inhibits Cell Cycle Progression in Human Breast Cancer Cells through Cdc2 Regulation. Cancer Res 66, 6615-6621 (2006).
Guagnini, F. et al. Neural contractions in colonic strips from patients with diverticular disease: role of endocannabinoids and substance P. Gut 55, 946-953 (2006).
Roberts, J. D., Gennings, C. & Shih, M. Synergistic affective analgesic interaction between delta-9-tetrahydrocannabinol and morphine. Eur J Pharmacol 530, 54-58 (2006).
Eichele, K., Weinzierl, U., Ramer, R., Brune, K. & Hinz, B. R(+)-methanandamide elicits a cyclooxygenase-2-dependent mitochondrial apoptosis signaling pathway in human neuroglioma cells. Pharm Res 23, 90-94 (2006).
Rao, G. K. & Kaminski, N. E. Induction of intracellular calcium elevation by Delta9-tetrahydrocannabinol in T cells involves TRPC1 channels. J Leukoc Biol 79, 202-213 (2006).
Ben-Shabat, S., Hanus, L. O., Katzavian, G. & Gallily, R. New cannabidiol derivatives: synthesis, binding to cannabinoid receptor, and evaluation of their antiinflammatory activity. J Med Chem 49, 1113-1117 (2006).
Grimaldi, C. et al. Anandamide inhibits adhesion and migration of breast cancer cells. Exp Cell Res 312, 363-373 (2006).
Vignot, S. et al. [Cannabis and cancer]. Bull Cancer 93, 163-170 (2006).
Shmist, Y. A. et al. Delta-9-tetrahydrocannabinol protects cardiac cells from hypoxia via CB2 receptor activation and nitric oxide production. Mol Cell Biochem 283, 75-83 (2006).
Hamamoto, D. T., Giridharagopalan, S. & Simone, D. A. Acute and chronic administration of the cannabinoid receptor agonist CP 55,940 attenuates tumor- evoked hyperalgesia. Eur J Pharmacol S (2006).
Pisanti, S. et al. Antiangiogenic activity of the endocannabinoid anandamide: Correlation to its tumor-suppressor efficacy. J Cell Physiol S (2006).
Ellert-Miklaszewska, A., Grajkowska, W., Gabrusiewicz, K., Kaminska, B. & Konarska, L. Distinctive pattern of cannabinoid receptor type II (CB2) expression in adult and pediatric brain tumors. Brain Res S (2006).
Huang, H., Nishi, K., Tsai, H. J. & Hammock, B. D. Development of highly sensitive fluorescent assays for fatty acid amide hydrolase. Anal Biochem SO – Anal Biochem. 2006 Dec 20;d (2006).
Davis, M., Maida, V., Daeninck, P. & Pergolizzi, J. The emerging role of cannabinoid neuromodulators in symptom management. Support Care Cancer (2006).
Wang, H. et al. Fatty acid amide hydrolase deficiency limits early pregnancy events. J Clin Invest 116, 2122-2131 (2006).
Jia, W. et al. Delta}9-Tetrahydrocannabinol-Induced Apoptosis in Jurkat Leukemia T Cells Is Regulated by Translocation of Bad to Mitochondria. Mol Cancer Res 4, 549-562 (2006).
Held-Feindt, J., Dorner, L., Sahan, G., Mehdorn, H. M. & Mentlein, R. Cannabinoid receptors in human astroglial tumors. J Neurochem 98, 886-893 (2006).
Perez, J. Combined cannabinoid therapy via an oromucosal spray. Drugs Today (Barc) 42, 495-503 (2006).
Matias, I. et al. Regulation, function, and dysregulation of endocannabinoids in models of adipose and beta-pancreatic cells and in obesity and hyperglycemia. J Clin Endocrinol Metab 91, 3171-3180 (2006).
Ben Amar, M. Cannabinoids in medicine: A review of their therapeutic potential. J Ethnopharmacol 105, 1-25 (2006).
Herrera, B. et al. The CB(2) cannabinoid receptor signals apoptosis via ceramide- dependent activation of the mitochondrial intrinsic pathway. Exp Cell Res (2006).
Holland, M. L. et al. The effects of cannabinoids on P-glycoprotein transport and expression in multidrug resistant cells. Biochem Pharmacol 71, 1146-1154 (2006).
Carracedo, A. et al. The stress-regulated protein p8 mediates cannabinoid-induced apoptosis of tumor cells. Cancer Cell 9, 301-312 (2006).
Verhoeckx, K. C. et al. Unheated Cannabis sativa extracts and its major compound THC-acid have potential immuno-modulating properties not mediated by CB1 and CB2 receptor coupled pathways. Int Immunopharmacol 6, 656-665 (2006).
Singh, J. & Budhiraja, S. Therapeutic potential of cannabinoid receptor ligands: current status. Methods Find Exp Clin Pharmacol 28, 177-183 (2006).
Russo, E. & Guy, G. W. A tale of two cannabinoids: the therapeutic rationale for combining tetrahydrocannabinol and cannabidiol. Med Hypotheses 66, 234-246 (2006).
Vidinsky, B., Gal, P. & Mojzis, J. [Different views on the association between cannabinoids and cancer]. Cas Lek Cesk 145, 453-7; discussion 458-9 (2006).
Massi, P., Vaccani, A. & Parolaro, D. Cannabinoids, immune system and cytokine network. Curr Pharm Des 12, 3135-3146 (2006).
Huskey, A. Cannabinoids in cancer pain management. J Pain Palliat Care Pharmacother 20, 43-46 (2006).
Melamede, R. J. Endocannabinoids: Multi-scaled, Global Homeostatic Regulators of Cells and Society. Interjournal Complex Systems 1669 (2006).
Sancho, R. et al. Mechanisms of HIV-1 inhibition by the lipid mediator N- arachidonoyldopamine. J Immunol 175, 3990-3999 (2005).
Melamede, R. Cannabis and tobacco smoke are not equally carcinogenic. Harm Reduct J 2, 21 (2005).
Sun, Y. X. et al. Involvement of N-acylethanolamine-hydrolyzing acid amidase in the degradation of anandamide and other N-acylethanolamines in macrophages. Biochim Biophys Acta 1736, 211-220 (2005).
Kogan, N. M. Cannabinoids and cancer. Mini Rev Med Chem 5, 941-952 (2005).
Sarnataro, D. et al. Plasma membrane and lysosomal localization of CB1 cannabinoid receptor are dependent on lipid rafts and regulated by anandamide in human breast cancer cells. FEBS Lett 579, 6343-6349 (2005).
Klein, T. W. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat Rev Immunol 5, 400-411 (2005).
McKallip, R. J., Nagarkatti, M. & Nagarkatti, P. S. Delta-9-tetrahydrocannabinol enhances breast cancer growth and metastasis by suppression of the antitumor immune response. J Immunol 174, 3281-3289 (2005).
Tashkin, D. P. Smoked marijuana as a cause of lung injury. Monaldi Arch Chest Dis 63, 93-100 (2005).
Bari, M., Paradisi, A., Pasquariello, N. & Maccarrone, M. Cholesterol-dependent modulation of type 1 cannabinoid receptors in nerve cells. J Neurosci Res 81, 275- 283 (2005).
Nithipatikom, K. et al. A new class of inhibitors of 2-arachidonoylglycerol hydrolysis and invasion of prostate cancer cells. Biochem Biophys Res Commun 332, 1028-1033 (2005).
Watanabe, K. et al. Marijuana extracts possess the effects like the endocrine disrupting chemicals. Toxicology 206, 471-478 (2005).
Sheng, W. S. et al. Synthetic cannabinoid WIN55,212-2 inhibits generation of inflammatory mediators by IL-1beta-stimulated human astrocytes. Glia 49, 211-219 (2005).
Ellert-Miklaszewska, A., Kaminska, B. & Konarska, L. Cannabinoids down- regulate PI3K/Akt and Erk signalling pathways and activate proapoptotic function of Bad protein. Cell Signal 17, 25-37 (2005).
Chung, K. F. Drugs to suppress cough. Expert Opin Investig Drugs 14, 19-27 (2005).
Hall, W., Christie, M. & Currow, D. Cannabinoids and cancer: causation, remediation, and palliation. Lancet Oncol 6, 35-42 (2005).
Ramirez, B. G., Blazquez, C., Gomez del Pulgar, T., Guzman, M. & de Ceballos, M. L. Prevention of Alzheimer’s disease pathology by cannabinoids: neuroprotection mediated by blockade of microglial activation. J Neurosci 25, 1904-1913 (2005).
Powles, T. et al. Cannabis-induced cytotoxicity in leukemic cell lines: the role of the cannabinoid receptors and the MAPK pathway. Blood 105, 1214-1221 (2005).
Radbruch, L. & Elsner, F. Emerging analgesics in cancer pain management. Expert Opin Emerg Drugs 10, 151-171 (2005).
Sharma, R., Tobin, P. & Clarke, S. J. Management of chemotherapy-induced nausea, vomiting, oral mucositis, and diarrhoea. Lancet Oncol 6, 93-102 (2005).
Roila, F. et al. Antiemetics in children receiving chemotherapy. Support Care Cancer 13, 129-131 (2005).
Kishimoto, S. et al. Endogenous cannabinoid receptor ligand induces the migration of human natural killer cells. J Biochem (Tokyo) 137, 217-223 (2005).
Flygare, J., Gustafsson, K., Kimby, E., Christensson, B. & Sander, B. Cannabinoid receptor ligands mediate growth inhibition and cell death in mantle cell lymphoma. FEBS Lett 579, 6885-6889 (2005).
Lee, S. Y., Oh, S. M., Lee, S. K. & Chung, K. H. Antiestrogenic effects of marijuana smoke condensate and cannabinoid compounds. Arch Pharm Res 28, 1365-1375 (2005).
Patsos, H. A., Hicks, D. J., Greenhough, A., Williams, A. C. & Paraskeva, C. Cannabinoids and cancer: potential for colorectal cancer therapy. Biochem Soc Trans 33, 712-714 (2005).
Ortega-Gutierrez, S. et al. Activation of the endocannabinoid system as therapeutic approach in a murine model of multiple sclerosis. FASEB J 19, 1338-1340 (2005).
Lombard, C., Nagarkatti, M. & Nagarkatti, P. S. Targeting cannabinoid receptors to treat leukemia: role of cross-talk between extrinsic and intrinsic pathways in Delta9-tetrahydrocannabinol (THC)-induced apoptosis of Jurkat cells. Leuk Res 29, 915-922 (2005).
McAllister, S. D. et al. Cannabinoids selectively inhibit proliferation and induce death of cultured human glioblastoma multiforme cells. J Neurooncol 74, 31-40 (2005).
Myrianthefs, P. M. & Batistaki, C. Cancer cachexia and immunomodulation. J BUON 10, 181-188 (2005).
Bari, M., Battista, N., Fezza, F., Finazzi-Agro, A. & Maccarrone, M. Lipid rafts control signaling of type-1 cannabinoid receptors in neuronal cells. Implications for anandamide-induced apoptosis. J Biol Chem 280, 12212-12220 (2005).
Fride, E., Bregman, T. & Kirkham, T. C. Endocannabinoids and food intake: newborn suckling and appetite regulation in adulthood. Exp Biol Med (Maywood) 230, 225-234 (2005).
Vaccani, A., Massi, P., Colombo, A., Rubino, T. & Parolaro, D. Cannabidiol inhibits human glioma cell migration through a cannabinoid receptor-independent mechanism. Br J Pharmacol 144, 1032-1036 (2005).
Guzman, M. Effects on cell viability. Handb Exp Pharmacol 627-642 (2005).
Izzo, A. A. & Coutts, A. A. Cannabinoids and the digestive tract. Handb Exp
Pharmacol 573-598 (2005).
Burstein, S. Ajulemic acid (IP-751): synthesis, proof of principle, toxicity studies,
and clinical trials. AAPS J 7, E143-8 (2005).
Carlini, E. A. The good and the bad effects of (-) trans-delta-9-tetrahydrocannabinol
(Delta 9-THC) on humans. Toxicon 44, 461-467 (2004).
Medveczky, M. M., Sherwood, T. A., Klein, T. W., Friedman, H. & Medveczky, P. G. Delta-9 tetrahydrocannabinol (THC) inhibits lytic replication of gamma oncogenic herpesviruses in vitro. BMC Med 2, 34 (2004).
Contassot, E. et al. Arachidonylethanolamide induces apoptosis of human glioma cells through vanilloid receptor-1. J Neuropathol Exp Neurol 63, 956-963 (2004).
Juttler, E. et al. The cannabinoid dexanabinol is an inhibitor of the nuclear factor- kappa B (NF-kappa B). Neuropharmacology 47, 580-592 (2004).
Velasco, G., Galve-Roperh, I., Sanchez, C., Blazquez, C. & Guzman, M. Hypothesis: cannabinoid therapy for the treatment of gliomas? Neuropharmacology 47, 315-323 (2004).
Inui, A. [Pathogenesis and treatment of cancer anorexia-cachexia, with special emphasis on aged patients]. Nippon Ronen Igakkai Zasshi 41, 460-467 (2004).
Bifulco, M. et al. A new strategy to block tumor growth by inhibiting endocannabinoid inactivation. FASEB J 18, 1606-1608 (2004).
Hinz, B., Ramer, R., Eichele, K., Weinzierl, U. & Brune, K. R(+)-methanandamide- induced cyclooxygenase-2 expression in H4 human neuroglioma cells: possible involvement of membrane lipid rafts. Biochem Biophys Res Commun 324, 621-626 (2004).
Rukwied, R., Gauter, B., Schley, M. & Konrad, C. [Cannabinoids-signal transduction and mode of action.]. Schmerz (2004).
Carter, G. T. & Ugalde, V. Medical marijuana: emerging applications for the management of neurologic disorders. Phys Med Rehabil Clin N Am 15, 943-54, ix (2004).
Fowler, C. J., Tiger, G., Ligresti, A., Lopez-Rodriguez, M. L. & Di Marzo, V. Selective inhibition of anandamide cellular uptake versus enzymatic hydrolysis–a difficult issue to handle. Eur J Pharmacol 492, 1-11 (2004).
Carter, G. T., Weydt, P., Kyashna-Tocha, M. & Abrams, D. I. Medicinal cannabis: rational guidelines for dosing. IDrugs 7, 464-470 (2004).
Matias, I. et al. Prostaglandin ethanolamides (prostamides): in vitro pharmacology and metabolism. J Pharmacol Exp Ther 309, 745-757 (2004).
Duran, M., Laporte, J. R. & Capella, D. [News about therapeutic use of Cannabis and endocannabinoid system]. Med Clin (Barc) 122, 390-398 (2004).
Hart, S., Fischer, O. M. & Ullrich, A. Cannabinoids induce cancer cell proliferation via tumor necrosis factor alpha-converting enzyme (TACE/ADAM17)-mediated transactivation of the epidermal growth factor receptor. Cancer Res 64, 1943-1950 (2004).
Massi, P. et al. Antitumor effects of cannabidiol, a nonpsychoactive cannabinoid, on human glioma cell lines. J Pharmacol Exp Ther 308, 838-845 (2004).
Carracedo, A. et al. Ceramide sensitizes astrocytes to oxidative stress: protective role of cannabinoids. Biochem J 380, 435-440 (2004).
Kraft, B. & Kress, H. G. [Cannabinoids and the immune system. Of men, mice and cells]. Schmerz 18, 203-210 (2004).
Nauck, F. & Klaschik, E. Cannabinoids in the treatment of the cachexia-anorexia syndrome in palliative care patients. Schmerz 18, 197-202 (2004).
Martin, B. R. & Wiley, J. L. Mechanism of action of cannabinoids: how it may lead to treatment of cachexia, emesis, and pain. J Support Oncol 2, 305-14; discussion 314-6 (2004).
Kogan, N. M. et al. Synthesis and antitumor activity of quinonoid derivatives of cannabinoids. J Med Chem 47, 3800-3806 (2004).
Derkinderen, P., Valjent, E., Darcel, F., Damier, P. & Girault, J. A. [Cannabis and cannabinoid receptors: from pathophysiology to therapeutic options]. Rev Neurol (Paris) 160, 639-649 (2004).
Rubovitch, V., Gafni, M. & Sarne, Y. The involvement of VEGF receptors and MAPK in the cannabinoid potentiation of Ca2+ flux into N18TG2 neuroblastoma cells. Brain Res Mol Brain Res 120, 138-144 (2004).
Desreumaux, P., Thuru, X. & Philippe, D. [Control of secretory diarrhea and colorectal cancer growth by cannabinoids in the gut]. Gastroenterol Clin Biol 28, 97 (2004).
Leggett, J. D. et al. Oleamide is a selective endogenous agonist of rat and human CB1 cannabinoid receptors. Br J Pharmacol 141, 253-262 (2004).
Sancho, R. et al. Immunosuppressive activity of endovanilloids: N-arachidonoyl- dopamine inhibits activation of the NF-kappa B, NFAT, and activator protein 1 signaling pathways. J Immunol 172, 2341-2351 (2004).
Cabral, G. A. & Marciano-Cabral, F. Cannabinoid-mediated exacerbation of brain infection by opportunistic amebae. J Neuroimmunol 147, 127-130 (2004).
Fowler, C. J. Possible involvement of the endocannabinoid system in the actions of three clinically used drugs. Trends Pharmacol Sci 25, 59-61 (2004).
Liang, Y. C., Huang, C. C. & Hsu, K. S. Therapeutic potential of cannabinoids in trigeminal neuralgia. Curr Drug Targets CNS Neurol Disord 3, 507-514 (2004).
Coutts, A. A. & Izzo, A. A. The gastrointestinal pharmacology of cannabinoids: an update. Curr Opin Pharmacol 4, 572-579 (2004).
Blazquez, C. et al. Cannabinoids inhibit the vascular endothelial growth factor pathway in gliomas. Cancer Res 64, 5617-5623 (2004).
Joseph, J., Niggemann, B., Zaenker, K. S. & Entschladen, F. Anandamide is an endogenous inhibitor for the migration of tumor cells and T lymphocytes. Cancer Immunol Immunother 53, 723-728 (2004).
Kalant, H. Adverse effects of cannabis on health: an update of the literature since 1996. Prog Neuropsychopharmacol Biol Psychiatry 28, 849-863 (2004).
Contassot, E., Tenan, M., Schnuriger, V., Pelte, M. F. & Dietrich, P. Y. Arachidonyl ethanolamide induces apoptosis of uterine cervix cancer cells via aberrantly expressed vanilloid receptor-1. Gynecol Oncol 93, 182-188 (2004).
Maccarrone, M. et al. The endocannabinoid system in human keratinocytes. Evidence that anandamide inhibits epidermal differentiation through CB1 receptor- dependent inhibition of protein kinase C, activation protein-1, and transglutaminase. J Biol Chem 278, 33896-33903 (2003).
Vannacci, A., Giannini, L., Masini, E. & Mannaioni, P. F. Cannabinoid-induced activation of ERK and AKT in mast cells may be mediated by intracellular NO production. J Immunol 171, 2767 (2003).
Fowler, C. J. et al. Inhibition of C6 glioma cell proliferation by anandamide, 1- arachidonoylglycerol, and by a water soluble phosphate ester of anandamide: variability in response and involvement of arachidonic acid. Biochem Pharmacol 66, 757-767 (2003).
Sanchez, M. G., Ruiz-Llorente, L., Sanchez, A. M. & Diaz-Laviada, I. Activation of phosphoinositide 3-kinase/PKB pathway by CB(1) and CB(2) cannabinoid receptors expressed in prostate PC-3 cells. Involvement in Raf-1 stimulation and NGF induction. Cell Signal 15, 851-859 (2003).
Portella, G. et al. Inhibitory effects of cannabinoid CB1 receptor stimulation on tumor growth and metastatic spreading: actions on signals involved in angiogenesis and metastasis. FASEB J 17, 1771-1773 (2003).
Ligresti, A. et al. Possible endocannabinoid control of colorectal cancer growth. Gastroenterology 125, 677-687 (2003).
Croci, T., Landi, M., Galzin, A. M. & Marini, P. Role of cannabinoid CB1 receptors and tumor necrosis factor-alpha in the gut and systemic anti-inflammatory activity of SR 141716 (rimonabant) in rodents. Br J Pharmacol 140, 115-122 (2003).
Klein, T. W. et al. The cannabinoid system and immune modulation. J Leukoc Biol 74, 486-496 (2003).
Guzman, M. Cannabinoids: potential anticancer agents. Nat Rev Cancer 3, 745-755 (2003).
Gardner, B., Zhu, L. X., Sharma, S., Tashkin, D. P. & Dubinett, S. M. Methanandamide increases COX-2 expression and tumor growth in murine lung cancer. FASEB J 17, 2157-2159 (2003).
Ramer, R., Weinzierl, U., Schwind, B., Brune, K. & Hinz, B. Ceramide is involved in r(+)-methanandamide-induced cyclooxygenase-2 expression in human neuroglioma cells. Mol Pharmacol 64, 1189-1198 (2003).
Wrobleski, S. T. et al. Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties. J Med Chem 46, 2110-2116 (2003).
Samson, M. T. et al. Differential roles of CB1 and CB2 cannabinoid receptors in mast cells. J Immunol 170, 4953-4962 (2003).
Kehl, L. J. et al. A cannabinoid agonist differentially attenuates deep tissue hyperalgesia in animal models of cancer and inflammatory muscle pain. Pain 103, 175-186 (2003).
Bifulco, M. & Di Marzo, V. [The endocannabinoid system as a target for the development of new drugs for cancer therapy]. Recenti Prog Med 94, 194-198 (2003).
Vandevoorde, S., Lambert, D. M., Smart, D., Jonsson, K. O. & Fowler, C. J. N- Morpholino- and N-diethyl-analogues of palmitoylethanolamide increase the sensitivity of transfected human vanilloid receptors to activation by anandamide without affecting fatty acid amidohydrolase activity. Bioorg Med Chem 11, 817-825 (2003).
Upham, B. L. et al. Cannabinoids inhibit gap junctional intercellular communication and activate ERK in a rat liver epithelial cell line. Int J Cancer 104, 12-18 (2003).
Kingsley, P. J. & Marnett, L. J. Analysis of endocannabinoids by Ag+ coordination tandem mass spectrometry. Anal Biochem 314, 8-15 (2003).
Blazquez, C. et al. Inhibition of tumor angiogenesis by cannabinoids. FASEB J 17, 529-531 (2003).
New toke on treating skin cancer. Lab Anim (NY) 32, 12 (2003).
Ware, M. A., Doyle, C. R., Woods, R., Lynch, M. E. & Clark, A. J. Cannabis use for chronic non-cancer pain: results of a prospective survey. Pain 102, 211-216 (2003).
Parker, L. A. et al. Effects of cannabinoids on lithium-induced conditioned rejection reactions in a rat model of nausea. Psychopharmacology (Berl) 166, 156-162 (2003).
Walsh, D., Nelson, K. A. & Mahmoud, F. A. Established and potential therapeutic applications of cannabinoids in oncology. Support Care Cancer 11, 137-143 (2003).
Bruera, E. & Castro, M. Cannabinoids in supportive care: are they necessary? Support Care Cancer 11, 133-134 (2003).
Tarzia, G. et al. Design, synthesis, and structure-activity relationships of alkylcarbamic acid aryl esters, a new class of fatty acid amide hydrolase inhibitors. J Med Chem 46, 2352-2360 (2003).
Klegeris, A., Bissonnette, C. J. & McGeer, P. L. Reduction of human monocytic cell neurotoxicity and cytokine secretion by ligands of the cannabinoid-type CB2 receptor. Br J Pharmacol 139, 775-786 (2003).
Bash, R., Rubovitch, V., Gafni, M. & Sarne, Y. The stimulatory effect of cannabinoids on calcium uptake is mediated by Gs GTP-binding proteins and cAMP formation. Neurosignals 12, 39-44 (2003).
Facchinetti, F., Del Giudice, E., Furegato, S., Passarotto, M. & Leon, A. Cannabinoids ablate release of TNFalpha in rat microglial cells stimulated with lypopolysaccharide. Glia 41, 161-168 (2003).
Ihenetu, K., Molleman, A., Parsons, M. E. & Whelan, C. J. Inhibition of interleukin-8 release in the human colonic epithelial cell line HT-29 by cannabinoids. Eur J Pharmacol 458, 207-215 (2003).
Casanova, M. L. et al. Inhibition of skin tumor growth and angiogenesis in vivo by activation of cannabinoid receptors. J Clin Invest 111, 43-50 (2003).
Zurier, R. B., Rossetti, R. G., Burstein, S. H. & Bidinger, B. Suppression of human monocyte interleukin-1beta production by ajulemic acid, a nonpsychoactive cannabinoid. Biochem Pharmacol 65, 649-655 (2003).
Sarafian, T. et al. Clarifying Cb2 Receptor-dependent and Independent Effects of Thc on Human Lung Epithelial Cells. Toxicol Appl Pharmacol (2008).
Davis, M. I., Ronesi, J. & Lovinger, D. M. A predominant role for inhibition of the adenylate cyclase/protein kinase A pathway in ERK activation by cannabinoid receptor 1 in N1E-115 neuroblastoma cells. J Biol Chem 278, 48973-48980 (2003).
Sanchez, M. G., Sanchez, A. M., Ruiz-Llorente, L. & Diaz-Laviada, I. Enhancement of androgen receptor expression induced by (R)-methanandamide in prostate LNCaP cells. FEBS Lett 555, 561-566 (2003).
Lavon, I. et al. A novel synthetic cannabinoid derivative inhibits inflammatory liver damage via negative cytokine regulation. Mol Pharmacol 64, 1334-1341 (2003).
Grotenhermen, F. & Muller-Vahl, K. IACM 2nd Conference on Cannabinoids in Medicine. Expert Opin Pharmacother 4, 2367-2371 (2003).
Keren, O. & Sarne, Y. Multiple mechanisms of CB1 cannabinoid receptors regulation. Brain Res 980, 197-205 (2003).
Kozak, K. R., Prusakiewicz, J. J., Rowlinson, S. W., Prudhomme, D. R. & Marnett, L. J. Amino acid determinants in cyclooxygenase-2 oxygenation of the endocannabinoid anandamide. Biochemistry 42, 9041-9049 (2003).
Gilgun-Sherki, Y., Melamed, E., Mechoulam, R. & Offen, D. The CB1 cannabinoid receptor agonist, HU-210, reduces levodopa-induced rotations in 6- hydroxydopamine-lesioned rats. Pharmacol Toxicol 93, 66-70 (2003).
Radbruch, L. & Nauck, F. [A review of side effects and complications with cannabinoid treatment]. Schmerz 17, 274-279 (2003).
Glaser, S. T. et al. Evidence against the presence of an anandamide transporter. Proc Natl Acad Sci U S A 100, 4269-4274 (2003).
Jonsson, K. O. et al. AM404 and VDM 11 non-specifically inhibit C6 glioma cell proliferation at concentrations used to block the cellular accumulation of the endocannabinoid anandamide. Arch Toxicol 77, 201-207 (2003).
Hinz, B., Ramer, R. & Brune, K. Induction of COX-2 expression by the endocannabinoid derivative R(+)-methanandamide. Adv Exp Med Biol 525, 145- 152 (2003).
Grotenhermen, F. Pharmacokinetics and pharmacodynamics of cannabinoids. Clin Pharmacokinet 42, 327-360 (2003).
Croxford, J. L. Therapeutic potential of cannabinoids in CNS disease. CNS Drugs 17, 179-202 (2003).
Reggio, P. H. Pharmacophores for ligand recognition and activation/inactivation of the cannabinoid receptors. Curr Pharm Des 9, 1607-1633 (2003).
Cannabis-based medicines–GW pharmaceuticals: high CBD, high THC, medicinal cannabis–GW pharmaceuticals, THC:CBD. Drugs R D 4, 306-309 (2003).
Gallily, R. et al. gamma-Irradiation Enhances Apoptosis Induced by Cannabidiol, a Non-psychotropic Cannabinoid, in Cultured HL-60 Myeloblastic Leukemia Cells. Leuk Lymphoma 44, 1767-1773 (2003).
Gomez Del Pulgar, T., De Ceballos, M. L., Guzman, M. & Velasco, G. Cannabinoids protect astrocytes from ceramide-induced apoptosis through the phosphatidylinositol 3-kinase/protein kinase B pathway. J Biol Chem 277, 36527- 36533 (2002).
Kozak, K. R. Metabolism of the endocannabinoids, 2-arachidonylglycerol and anandamide, into prostaglandin, thromboxane, and prostacyclin glycerol esters and ethanolamides. J Biol Chem 277, 44877-44885 (2002).
Hashibe, M., Ford, D. E. & Zhang, Z. F. Marijuana smoking and head and neck cancer. J Clin Pharmacol 42, 103S-107S (2002).
Rubovitch, V., Gafni, M. & Sarne, Y.
http://weedbay.net/index.php/component/content/article/1974
ADD/ ADHD
Marijuana and ADD Therapeutic uses of Medical Marijuana in the treatment of ADD
http://www.onlinepot.org/medical/add&mmj.htm
Cannabis as a medical treatment for attention deficit disorder
http://www.chanvre-info.ch/info/en/…-treatment.html
Cannabinoids effective in animal model of hyperactivity disorder
http://www.cannabis-med.org/english…el.php?id=162#4
Cannabis ‘Scrips to Calm Kids?
http://www.foxnews.com/story/0,2933,117541,00.html
Addiction risk- Physical
Women’s Guide to the UofC
http://wguide.uchicago.edu/9substance.html
Cannabis Basics
http://www.erowid.org/plants/cannab…is_basics.shtml
10 Things Every Parent, Teenager & Teacher Should Know About Marijuana (4th Q)
http://www.erowid.org/plants/cannab…is_flyer1.shtml
Marijuana Myths, Claim No. 9
http://www.erowid.org/plants/cannab…bis_myth9.shtml
AIDS – see HIV
Alcoholism
Role of cannabinoid receptors in alcohol abuse
http://www.medicalnewstoday.com/articles/30338.php
Cannabidiol, Antioxidants, and Diuretics in Reversing Binge Ethanol-Induced Neurotoxicity
http://jpet.aspetjournals.org/cgi/c…ourcetype=HWCIT
Cannabis substitution
http://www.cannabis-med.org/studies…how.php?s_id=86
Cannabis as a Substitute for Alcohol
http://ccrmg.org/journal/03sum/substitutealcohol.html
ALS
Cannabinol delays symptom onset
http://www.ncbi.nlm.nih.gov/sites/e…t_uids=16183560
Marijuana in the management of amyotrophic lateral sclerosis
http://www.medscape.com/medline/abstract/11467101
Cannabis use in patients with amyotrophic lateral sclerosis.
http://www.medscape.com/medline/abstract/15055508
Cannabis Relieves Lou Gehrigs Symptoms
http://www.rense.com/general51/lou.htm
Cannabis’ Potential Exciting Researchers in Treatment of ALS, Parkinson’s Disease
http://66.218.69.11/search/cache?ei…&icp=1&.intl=us
Alzheimers
MARIJUANA SLOWS ALZHEIMER’S DECLINE
http://www.mapinc.org/drugnews/v05/n307/a10.html
Marijuana may block Alzheimer’s
http://news.bbc.co.uk/2/hi/health/4286435.stm
Prevention of Alzheimer’s Disease Pathology by Cannabinoids
http://www.jneurosci.org/cgi/content/abstract/25/8/1904
Marijuana’s Active Ingredient Shown to Inhibit Primary Marker of Alzheimer’s Disease
http://www.pacifier.com/~alive/articles/ca060809.htm
Dronabinol in the treatment of agitation in patients with Alzheimer’s disease with anorexia
http://www.cannabis-med.org/studies…how.php?s_id=61
Dronabinol in the treatment of refractory agitation in Alzheimer’s disease
http://www.cannabis-med.org/studies…how.php?s_id=92
Effects of dronabinol on anorexia and disturbed behavior in patients with Alzheimer’s disease.
http://www.cannabis-med.org/studies…how.php?s_id=59
Cannabinoids reduce the progression of Alzheimer’s disease in animals
http://www.cannabis-med.org/english…el.php?id=187#1
Molecular Link between the Active Component of Marijuana and Alzheimer’s Disease Pathology
http://www.unboundmedicine.com/medl…sease_Pathology
THC inhibits primary marker of Alzheimer’s disease
http://www.cannabis-med.org/english…el.php?id=225#3
——— Page 1
Amotivational Syndrome
Amotivational Syndrome
http://leda.lycaeum.org/?ID=12454
Marijuana Myths, Claim No. 11
http://www.erowid.org/plants/cannab…is_myth11.shtml
Debunking ‘Amotivational Syndrome’
http://www.mapinc.org/drugnews/v06/n400/a06.html
Amotivational Syndrome
http://www.bookrags.com/Amotivational_syndrome
Debunking the Amotivational Syndrome
http://www.drugscience.org/Petition/C3F.html
Cannabis Use Not Linked To So-Called “Amotivational Syndrome”
http://www.norml.org/index.cfm?Grou…tm_format=print
Anecdotal Evidence/First person stories
Shared Comments and Observations
http://www.rxmarihuana.com/comments…bservations.htm
Cannabis Sativa (Marijuana) for Fibromyalgia
http://www.fibromyalgia-reviews.com/Drg_Marijuana.cfm
ANECDOTAL ARTICLES
http://cannabislink.ca/medical/#medanecdotal
Testimonials
http://www.benefitsofmarijuana.com/testimonials.html
Excerpts of testimonials.
http://www.ganjaland.com/freemedicalseeds.htm
Appetite Stimulant
Dronabinol an effective appetite stimulant?
http://www.cannabis-med.org/studies…ow.php?s_id=188
THC improves appetite and reverses weight loss in AIDS patients
http://www.cannabis-med.org/studies…ow.php?s_id=189
Efficacy of dronabinol alone and in combination
http://www.cannabis-med.org/studies…ow.php?s_id=191
Dronabinol and marijuana in HIV-positive marijuana smokers: caloric intake, mood, and sleep.
http://www.cannabis-med.org/studies…ow.php?s_id=190
The synthetic cannabinoid nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases
http://www.cannabis-med.org/studies…ow.php?s_id=180
Safety and efficacy of dronabinol in the treatment of agitation in patients with Alzheimer’s disease
http://www.cannabis-med.org/studies…how.php?s_id=61
The perceived effects of smoked cannabis on patients with multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=13
Effects of dronabinol on anorexia and disturbed behavior in patients with Alzheimer’s disease
http://www.cannabis-med.org/studies…how.php?s_id=59
Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS.
http://www.cannabis-med.org/studies…how.php?s_id=21
Delta-9-tetrahydrocannabinol for appetite stimulation in cancer-associated anorexia
http://www.cannabis-med.org/studies…how.php?s_id=52
Effect of dronabinol on nutritional status in HIV infection.
http://www.cannabis-med.org/studies…ow.php?s_id=150
Dronabinol stimulates appetite and causes weight gain in HIV patients.
http://www.cannabis-med.org/studies…how.php?s_id=20
Dronabinol effects on weight in patients with HIV infection.
http://www.cannabis-med.org/studies…how.php?s_id=45
Recent clinical experience with dronabinol.
http://www.cannabis-med.org/studies…how.php?s_id=90
Dronabinol enhancement of appetite in cancer patients.
http://www.cannabis-med.org/studies…ow.php?s_id=149
Effects of smoked marijuana on food intake and body weight
http://www.cannabis-med.org/studies…ow.php?s_id=117
Behavioral analysis of marijuana effects on food intake in humans.
http://www.cannabis-med.org/studies…ow.php?s_id=118
Cancer-related anorexia-cachexia syndrome
http://www.unboundmedicine.com/medl…xia_Study_Group
THC effective in appetite and weight loss in severe lung disease (COPD)
http://www.cannabis-med.org/english…el.php?id=191#2
Machinery Of The ‘Marijuana Munchies’
http://www.sciencedaily.com/release…51226102503.htm
Arthritis
Cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis
http://www.pnas.org/cgi/content/full/97/17/9561
The Cannabinergic System as a Target for Anti-inflammatory Therapies
http://www.ingentaconnect.com/conte…000013/art00008
Sativex in the treatment of pain caused by rheumatoid arthritis
http://rheumatology.oxfordjournals….bstract/45/1/50
Suppression of fibroblast metalloproteinases by ajulemic acid,
http://ccicnewsletter.com/index.php…06_Rheumatology
The antinociceptive effect of Delta9-tetrahydrocannabinol in the arthritic rat
http://www.unboundmedicine.com/medl…binoid_receptor
Synergy between Delta(9)-tetrahydrocannabinol and morphine in the arthritic rat
http://www.unboundmedicine.com/medl…e_arthritic_rat
Cannabis based medicine eases pain and suppresses disease
http://www.medicalnewstoday.com/articles/33376.php
Pot-Based Drug Promising for Arthritis
http://www.webmd.com/rheumatoid-art…g-for-arthritis
Asthma
The Cannabinergic System as a Target for Anti-inflammatory Therapies
http://www.ingentaconnect.com/conte…000013/art00008
Acute and subacute bronchial effects of oral cannabinoids.
http://www.cannabis-med.org/studies…how.php?s_id=44
Comparison of bronchial effects of nabilone and terbutaline
http://www.cannabis-med.org/studies…how.php?s_id=43
Bronchial effects of aerosolized delta 9-tetrahydrocannabinol
http://www.cannabis-med.org/studies…ow.php?s_id=109
Bronchodilator effect of delta1-tetrahydrocannabinol administered by aerosol
http://www.cannabis-med.org/studies…how.php?s_id=60
Effects of smoked marijuana in experimentally induced asthma.
http://www.cannabis-med.org/studies…how.php?s_id=57
Marijuana and oral delta9-tetrahydrocannabinol on specific airway conductance
http://www.cannabis-med.org/studies…how.php?s_id=67
New Synthetic Delta-9-THC Inhaler Offers Safe, Rapid Delivery
http://www.medicalnewstoday.com/articles/22937.php
Smoked marijuana and oral delta-9-THC on specific airway conductance in asthmatic subjects
http://www.ukcia.org/research/Smoke…InAsthmatic.php
Atherosclerosis
Marijuana Chemical Fights Hardened Arteries
http://www.webmd.com/heart-disease/…rdened-arteries
Does Cannabis Hold the Key to Treating Cardiometabolic Disease
http://www.medscape.com/viewarticle/525040_print
Cannabis may keep arteries clear
http://www.gnn.tv/headlines/2634/Ca…_arteries_clear
The Cannabinergic System as a Target for Anti-inflammatory Therapies
http://www.ingentaconnect.com/conte…000013/art00008
Cannabis compound tackles blood vessel disease
http://www.medicalnewstoday.com/articles/22658.php
Medical marijuana: study shows that THC slows atherosclerosis
http://thenexthurrah.typepad.com/th…al_marijua.html
Cardiovascular Effects of Cannabis
http://www.idmu.co.uk/canncardio.htm
Atrophie Blanche
Atrophie Blanche Treated With Cannabis and/or THC
http://ccrmg.org/journal/04spr/clinical.html#thm
Autism
Autism and Medical Marijuana
http://www.autism.org/marijuana.html
THE SAM PROJECT: James D.
http://www.letfreedomgrow.com/articles/james_d.htm
Medical marijuana: a valuable treatment for autism?
http://www.autismwebsite.com/ari/ne…r/marijuana.htm
——— Page 2
Cancer – breast
Anandamide inhibits human breast cancer cell proliferation
http://www.pnas.org/cgi/content/abstract/95/14/8375
Inhibition of Human Breast and Prostate Cancer Cell Proliferation1
http://endo.endojournals.org/cgi/co…tract/141/1/118
Antitumor Activity of Plant Cannabinoids
http://jpet.aspetjournals.org/cgi/c…ract/318/3/1375
9-Tetrahydrocannabinol Inhibits Cell Cycle Progression in Human Breast Cancer
http://cancerres.aacrjournals.org/c…ract/66/13/6615
Cannabidiol inhibits tumour growth in leukaemia and breast cancer
http://www.cannabis-med.org/english…el.php?id=220#2
THC and prochlorperazine effective in reducing vomiting in women following breast surgery
http://www.cannabis-med.org/english…el.php?id=219#1
Cancer- colorectal
Anandamide, induces cell death in colorectal carcinoma cells
http://gut.bmj.com/cgi/content/abstract/54/12/1741
Cannabinoids and cancer: potential for colorectal cancer therapy.
http://www.medscape.com/medline/abstract/16042581
Cancer- glioma/ brain
Anti-tumor effects of cannabidiol
http://www.hempworld.com/HempPharm/…milanstudy.html
Pot’s cancer healing properties
http://www.november.org/stayinfo/br…ncerKiller.html
Cannabinoids Inhibit the Vascular Endothelial Growth Factor Pathway in Gliomas
http://cancerres.aacrjournals.org/c…hort/64/16/5617
Inhibition of Glioma Growth in Vivo
http://cancerres.aacrjournals.org/c…/61/15/5784.pdf
Delta(9)-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme.
http://www.cannabis-med.org/studies…ow.php?s_id=193
Cannabidiol triggers caspase activation and oxidative stress in human glioma cells.
http://www.ihop-net.org/UniPub/iHOP…l?pmid=16909207
Cannabinoid receptors in human astroglial tumors
http://www.brainlife.org/abstracts/…t_j20060800.pdf
Cannabis extract makes brain tumors shrink, halts growth of blood vessels
http://www.medicalnewstoday.com/articles/12088.php
THC tested against brain tumour in pilot clinical study
http://www.cannabis-med.org/english…el.php?id=222#1
Cancer- leukemia
Cannabis-induced cytotoxicity in leukemic cell lines
http://bloodjournal.hematologylibra…ract/105/3/1214
Cannabidiol-Induced Apoptosis in Human Leukemia Cells
http://molpharm.aspetjournals.org/c…stract/70/3/897
Marijuana’s Active Ingredient Kills Leukemia Cells
http://www.treatingyourself.com/vbu…read.php?t=7107
Targeting CB2 cannabinoid receptors to treat malignant lymphoblastic disease
http://bloodjournal.hematologylibra…t/100/2/627.pdf
Cannabinoids induce incomplete maturation of cultured human leukemia cells
http://www.osti.gov/energycitations…osti_id=5164483
{Delta}9-Tetrahydrocannabinol-Induced Apoptosis in Jurkat Leukemia T Cells
http://mcr.aacrjournals.org/cgi/con…bstract/4/8/549
Cannabidiol inhibits tumour growth in leukaemia and breast cancer
http://www.cannabis-med.org/english…el.php?id=220#2
Cancer- lung
Antineoplastic activity of cannabinoids
http://www.ukcia.org/research/Antin…ds/default.html
Delta(9)-Tetrahydrocannabinol inhibits epithelial growth factor-induced lung cancer cell migration
http://www.unboundmedicine.com/medl…astasis_in_vivo
Smoking Cannabis Does Not Cause Cancer Of Lung or Upper Airways
http://ccrmg.org/journal/05aut/nocancer.html
No association between lung cancer and cannabis smoking in large study
http://www.cannabis-med.org/english…el.php?id=219#2
Marijuana Smoking Found Non-Carcinogenic
http://www.medpagetoday.com/Hematol…gCancer/tb/3393
CLAIM #4: MARIJUANA CAUSES LUNG DISEASE
http://www.erowid.org/plants/cannab…bis_myth4.shtml
Cancer- melanoma
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases.
http://www.cannabis-med.org/studies…ow.php?s_id=180
Intractable nausea and vomiting due to gastrointestinal mucosal metastases
http://www.cannabis-med.org/studies…how.php?s_id=35
Cancer – oral
Smoking of cannabis does not increase risk for oral cancer
http://www.cannabis-med.org/english…el.php?id=175#1
Marijuana use and Risk of Oral Squamous Cell Carcinoma
http://66.218.69.11/search/cache?ei…&icp=1&.intl=us
Cancer-pancreatic
Cannabinoids Induce Apoptosis of Pancreatic Tumor Cells
http://cancerres.aacrjournals.org/c…ract/66/13/6748
Cancer – prostate
Inhibition of Human Breast and Prostate Cancer Cell Proliferation
http://endo.endojournals.org/cgi/co…tract/141/1/118
Cannabinoid Receptor as a Novel Target for the Treatment of Prostate Cancer
http://cancerres.aacrjournals.org/c…t/65/5/1635.pdf
——- Page 3
Cancer – Risk Cannabis vs Tobacco
Cannabis Smoke and Cancer: Assessing the Risk
http://www.norml.org/index.cfm?Group_ID=6891
Cannabis and tobacco smoke are not equally carcinogenic
http://www.pubmedcentral.nih.gov/ar…i?artid=1277837
Smoking Marijuana Does Not Cause Lung Cancer
http://www.mapinc.org/drugnews/v05/n1065/a03.html
Blunt Smokers Link Dependence Potential To Nicotine
http://www.medicalnewstoday.com/articles/52838.php
Premiere British Medical Journal Pronounces Marijuana Safer Than Alcohol, Tobacco
http://cannabislink.ca/medical/safer.html
Why Doesn’t Smoking Marijuana Cause Cancer?
http://www.healthcentral.com/drdean/408/14275.html
Marijuana Smoking Found Non-Carcinogenic
http://www.medpagetoday.com/Hematol…gCancer/tb/3393
Cancer – Skin
Inhibition of skin tumor growth
http://www.jci.org/cgi/content/full…y=MpUgjDbqHybAU
Cannabis Reduces Skin Cancer
http://www.onlinepot.org/medical/skincancerreport.htm
Cancer – Testicular
The antiemetic efficacy of nabilone
http://www.cannabis-med.org/studies…ow.php?s_id=127
Chemotherapy for Testicular Cancer
http://www.rxmarihuana.com/shared_c…icularchemo.htm
Cancer –various/ unnamed
Derivatives of cannabis for anti-cancer treatment
http://www.eurekalert.org/pub_relea…uo-do060605.php
Cancer Killer
http://www.november.org/stayinfo/br…ncerKiller.html
Anandamide Induces Apoptosis
http://www.jbc.org/cgi/content/abstract/275/41/31938
Nabilone improves pain and symptom management
http://www.cannabis-med.org/studies…ow.php?s_id=177
The effects of smoked cannabis in painful peripheral neuropathy
http://www.cannabis-med.org/studies…how.php?s_id=96
Delta-9-tetrahydrocannabinol for appetite stimulation
http://www.cannabis-med.org/studies…how.php?s_id=52
Dronabinol and prochlorperazine in combination
http://www.cannabis-med.org/studies…how.php?s_id=28
Dronabinol enhancement of appetite in cancer patients.
http://www.cannabis-med.org/studies…ow.php?s_id=149
Efficacy of tetrahydrocannabinol
http://www.cannabis-med.org/studies…how.php?s_id=31
Inhalation marijuana as an antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=155
Nabilone versus domperidone
http://www.cannabis-med.org/studies…ow.php?s_id=129
Inhalation marijuana as an antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=155
Nabilone vs. placebo in chemotherapy-induced nausea and vomiting
http://www.cannabis-med.org/studies…ow.php?s_id=156
The antiemetic activity of tetrahydrocanabinol versus metoclopramide
http://www.cannabis-med.org/studies…how.php?s_id=24
Delta-9-tetrahydrocannabinol as an antiemetic for patients receiving cancer chemotherapy
http://www.cannabis-med.org/studies…show.php?s_id=5
Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate
http://www.cannabis-med.org/studies…how.php?s_id=23
Delta-9-tetrahydrocannabinol (THC) as an antiemetic in patients treated with cancer chemotherapy
http://www.cannabis-med.org/studies…how.php?s_id=27
Amelioration of cancer chemotherapy-induced nausea and vomiting by delta-9-THC
http://www.cannabis-med.org/studies…ow.php?s_id=107
Superiority of nabilone over prochlorperazine as an antiemetic
http://www.cannabis-med.org/studies…ow.php?s_id=126
Analgesic effect of delta-9-tetrahydrocannabinol.
http://www.cannabis-med.org/studies…how.php?s_id=16
The analgesic properties of delta-9-tetrahydrocannabinol and codeine.
http://www.cannabis-med.org/studies…how.php?s_id=17
Comparison of orally administered cannabis extract and delta-9-THC
http://www.unboundmedicine.com/medl…xia_Study_Group
Cannabis May Help Combat Cancer-causing Herpes Viruses
http://www.sciencedaily.com/release…40923092627.htm
Marijuana Smoking Found Non-Carcinogenic
http://www.medpagetoday.com/Hematol…gCancer/tb/3393
Cannabidiol
Cannabidiol, Antioxidants, and Diuretics in Reversing Binge Ethanol-Induced Neurotoxicity
http://jpet.aspetjournals.org/cgi/c…ourcetype=HWCIT
Cannabinol delays symptom onset
http://www.ncbi.nlm.nih.gov/sites/e…t_uids=16183560
Cannabidiol is an oral anti-arthritic therapeutic in murine collagen-induced arthritis
http://www.pnas.org/cgi/content/full/97/17/9561
Cannabidiol inhibits tumour growth in leukaemia and breast cancer
http://www.cannabis-med.org/english…el.php?id=220#2
Anti-tumor effects of cannabidiol
http://www.hempworld.com/HempPharm/…milanstudy.html
Cannabidiol triggers caspase activation and oxidative stress in human glioma cells.
http://www.ihop-net.org/UniPub/iHOP…l?pmid=16909207
Cannabidiol-Induced Apoptosis in Human Leukemia Cells
http://molpharm.aspetjournals.org/c…stract/70/3/897
Cannabidiol inhibits tumour growth in leukaemia and breast cancer
http://www.cannabis-med.org/english…el.php?id=220#2
Cannabidiol lowers incidence of diabetes in non-obese diabetic mice
http://www.ingentaconnect.com/conte…sn7o5efqr.alice
Neuroprotective and Blood-Retinal Barrier-Preserving Effects of Cannabidiol
http://ajp.amjpathol.org/cgi/content/full/168/1/235
Evaluation of cannabidiol in dystonic movement disorders
http://www.cannabis-med.org/studies…how.php?s_id=14
Cannabidiol in dystonic movement disorders.
http://www.cannabis-med.org/studies…ow.php?s_id=139
Beneficial and adverse effects of cannabidiol in a Parkinson patient
http://www.cannabis-med.org/studies…ow.php?s_id=142
Treatment of Meige’s syndrome with cannabidiol.
http://www.cannabis-med.org/studies…ow.php?s_id=114
CANNABIDIOL TO HEALTHY VOLUNTEERS AND EPILEPTIC PATIENTS
http://web.acsalaska.net/~warmgun/es201.html
Chronic administration of cannabidiol to healthy volunteers and epileptic patients.
http://www.cannabis-med.org/studies…how.php?s_id=42
Neuroprotective effect of (-)Delta9-tetrahydrocannabinol and cannabidiol
http://www.unboundmedicine.com/medl…f_peroxynitrite
EFFECTS OF CANNABIDIOL IN HUNTINGTON’S DISEASE
http://www.druglibrary.org/schaffer…al/hunting1.htm
The therapeutic rationale for combining tetrahydrocannabinol and cannabidiol.
http://www.medscape.com/medline/abstract/16209908
Cannabidiol has a cerebroprotective action
http://www.unboundmedicine.com/medl…iting_mechanism
Cannabidiol as an antipsychotic
http://www.cannabis-med.org/studies…ow.php?s_id=171
Cannabidiol, a constituent of Cannabis sativa, modulates sleep in rats.
http://www.medscape.com/medline/abs…844117?prt=true
Who’s Afraid of Cannabidiol?
http://www.counterpunch.org/gardner07142007.html
Chemical composition
Cannabis: A source of useful pharma compounds
http://www.medpot.net/forums/index.php?showtopic=18608
Pharmacokinetics and cannabinoid action using oral cannabis extract
http://www.pharma-lexicon.com/medic…hp?newsid=29638
Pharmacokinetics of cannabinoids
http://66.218.69.11/search/cache?ei…&icp=1&.intl=us
The chemistry and biological activity of cannabis
http://www.unodc.org/unodc/en/bulle….html?print=yes
Differential effects of medical marijuana based on strain and route of administration
http://www.medicalmarijuanaprocon.o…trainsstudy.pdf
What is THC?
http://www.medicalmarijuanaprocon.o…1.0373456855945
Cannabis / Marijuana ( ? 9 -Tetrahydrocannabinol, THC)
http://www.nhtsa.dot.gov/people/inj…gs/cannabis.htm
———- Page 4
Chemotherapy
Efficacy of dronabinol alone and in combination
http://www.cannabis-med.org/studies…ow.php?s_id=191
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases
http://www.cannabis-med.org/studies…ow.php?s_id=180
Intractable nausea and vomiting
http://www.cannabis-med.org/studies…how.php?s_id=35
An efficient new cannabinoid antiemetic in pediatric oncology
http://www.cannabis-med.org/studies…show.php?s_id=7
Dronabinol and prochlorperazine in combination
http://www.cannabis-med.org/studies…how.php?s_id=28
Marijuana as antiemetic medicine
http://www.cannabis-med.org/studies…ow.php?s_id=134
Efficacy of tetrahydrocannabinol in patients refractory to standard anti-emetic therapy
http://www.cannabis-med.org/studies…how.php?s_id=31
Inhalation marijuana as an antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=155
Nabilone versus prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=120
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Antiemetic efficacy of nabilone and alizapride
http://www.cannabis-med.org/studies…ow.php?s_id=127
Nabilone versus domperidone
http://www.cannabis-med.org/studies…ow.php?s_id=129
THC or Compazine for the cancer chemotherapy patient
http://www.cannabis-med.org/studies…how.php?s_id=34
Comparison of nabilone and prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=128
Nabilone vs. prochlorperazine for refractory emesis
http://www.cannabis-med.org/studies…ow.php?s_id=146
Nabilone vs. placebo
http://www.cannabis-med.org/studies…ow.php?s_id=156
Tetrahydroannabinol (THC) vs prochlorperazine as chemotherapy antiemetics.
http://www.cannabis-med.org/studies…how.php?s_id=30
Comparative trial of the antiemetic effects of THC and haloperidol
http://www.cannabis-med.org/studies…how.php?s_id=64
Comparison of delta-9-tetrahydrocannabinol and prochlorperazine
http://www.cannabis-med.org/studies…show.php?s_id=3
Delta 9-tetrahydrocannabinol in cancer chemotherapy.
http://www.cannabis-med.org/studies…how.php?s_id=88
Antiemetic effect of tetrahydrocannabinol
http://www.cannabis-med.org/studies…show.php?s_id=6
Tetrahydrocanabinol versus metoclopramide and thiethylperazine
http://www.cannabis-med.org/studies…how.php?s_id=24
Effects of nabilone and prochlorperazine on chemotherapy-induced emesis
http://www.cannabis-med.org/studies…ow.php?s_id=131
Delta-9-tetrahydrocannabinol as an antiemetic
http://www.cannabis-med.org/studies…show.php?s_id=5
Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate
http://www.cannabis-med.org/studies…how.php?s_id=23
THC as an antiemetic in patients treated with cancer chemotherapy
http://www.cannabis-med.org/studies…how.php?s_id=27
Amelioration of cancer chemotherapy-induced nausea and vomiting by delta-9-THC
http://www.cannabis-med.org/studies…ow.php?s_id=107
Superiority of nabilone over prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=126
Antiemetic effect of delta-9-tetrahydrocannabinol
http://www.cannabis-med.org/studies…show.php?s_id=4
Children
Experiences with THC-treatment in children and adolescents
http://www.cannabis-med.org/studies…how.php?s_id=80
An efficient new cannabinoid antiemetic in pediatric oncology.
http://www.cannabis-med.org/studies…show.php?s_id=7
Nabilone versus prochlorperazine for control of cancer chemotherapy-induced emesis in children
http://www.cannabis-med.org/studies…ow.php?s_id=120
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Marijuana and ADD Therapeutic uses of Medical Marijuana in the treatment of ADD
http://www.onlinepot.org/medical/add&mmj.htm
Oily fish makes ‘babies brainier’
http://news.bbc.co.uk/2/hi/health/4631006.stm
Cannabis is a First-Line Treatment for Childhood Mental Disorders
http://www.counterpunch.org/mikuriya07082006.html
Ganja use among Jamaican women.
http://www.rism.org/isg/dlp/ganja/a…anjaBabyes.html
Dreher’s Jamaican Pregnancy Study
http://www.november.org/stayinfo/br…reherStudy.html
Cannabis Relieves Morning Sickness
http://ccrmg.org/journal/06spr/dreher.html#morning
Moderate cannabis use not harmful to the brain of adolescents, M R I study finds
http://www.cannabis-med.org/english…el.php?id=218#3
No brain structural change associated with adolescent cannabis use
http://www.pubmedcentral.nih.gov/bo…l&artid=1524733
No ‘Smoking’ Gun: Research Indicates Teen Marijuana Use Does Not Predict Drug, Alcohol Abuse
http://www.sciencedaily.com/release…61204123422.htm
Pot May Not Shrink Teens’ Brains After All
http://www.medpagetoday.com/Neurolo…urology/tb/3242
Chronic Cystitis
Cannabinoid rotation in a young woman with chronic cystitis
http://www.cannabis-med.org/studies…ow.php?s_id=115
CPOD
THC effective in appetite and weight loss in severe lung disease (COPD)
http://www.cannabis-med.org/english…el.php?id=191#2
Heavy Long-Term Marijuana Use Does Not Impair Lung Function
http://www.erowid.org/plants/cannab…is_media7.shtml
Diabetes
Cannabinoid Reduces Incidence Of Diabetes
http://www.norml.org/index.cfm?Group_ID=6909
Marijuana Compound May Help Stop Diabetic Retinopathy
http://www.sciencedaily.com/release…60227184647.htm
Cannabidiol lowers incidence of diabetes in non-obese diabetic mice
http://www.ingentaconnect.com/conte…sn7o5efqr.alice
Anticoagulant Effects of a Cannabis Extract in an Obese Rat Model
http://www.level1diet.com/research/id/14687
Neuroprotective and Blood-Retinal Barrier-Preserving Effects of Cannabidiol
http://ajp.amjpathol.org/cgi/content/full/168/1/235
The Cannabinergic System as a Target for Anti-inflammatory Therapies
http://www.ingentaconnect.com/conte…000013/art00008
Effect of tetrahydrocurcumin on blood glucose, plasma insulin and hepatic key enzymes
http://www.unboundmedicine.com/medl…d_diabetic_rats
Cannabidiol reduces the development of diabetes in an animal study
http://www.cannabis-med.org/english…el.php?id=219#3
Depression
Cannabinoids promote hippocampus neurogenesis and produce anxiolytic- and antidepressant
http://www.jci.org/cgi/content/full/115/11/3104
Antidepressant-like activity by blockade of anandamide hydrolysis
http://www.pubmedcentral.nih.gov/ar…bmedid=16352709
Decreased depression in marijuana users.
http://www.medscape.com/medline/abstract/15964704
Antidepressant-like activity
http://www.pubmedcentral.nih.gov/ar…bmedid=16352709
Dronabinol and marijuana in HIV-positive marijuana smokers: caloric intake, mood, and sleep.
http://www.cannabis-med.org/studies…ow.php?s_id=190
Nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
The perceived effects of smoked cannabis on patients with multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=13
Cannabis and Depression
http://www.pacifier.com/~alive/cmu/…nd_cannabis.htm
Association between cannabis use and depression may not be causal, study says
http://www.cannabis-med.org/english…el.php?id=177#4
Marijuana use and depression among adults: Testing for causal associations.
http://www.ncbi.nlm.nih.gov/sites/e…Pubmed_RVDocSum
Do patients use marijuana as an antidepressant?
http://www.ncbi.nlm.nih.gov/sites/e…Pubmed_RVDocSum
Dermatitis
Efficacy of dietary hempseed oil in patients with atopic dermatitis.
http://www.medscape.com/medline/abs…ryText=hempseed
Dronabinol
Dronabinol in the treatment of agitation in patients with Alzheimer’s disease with anorexia
http://www.cannabis-med.org/studies…how.php?s_id=61
Dronabinol in the treatment of refractory agitation in Alzheimer’s disease
http://www.cannabis-med.org/studies…how.php?s_id=92
Effects of dronabinol on anorexia and disturbed behavior in patients with Alzheimer’s disease.
http://www.cannabis-med.org/studies…how.php?s_id=59
Dronabinol an effective appetite stimulant?
http://www.cannabis-med.org/studies…ow.php?s_id=188
Safety and efficacy of dronabinol in the treatment of agitation in patients with Alzheimer’s disease
http://www.cannabis-med.org/studies…how.php?s_id=61
Effect of dronabinol on nutritional status in HIV infection.
http://www.cannabis-med.org/studies…ow.php?s_id=150
Dronabinol stimulates appetite and causes weight gain in HIV patients.
http://www.cannabis-med.org/studies…how.php?s_id=20
Dronabinol effects on weight in patients with HIV infection.
http://www.cannabis-med.org/studies…how.php?s_id=45
Recent clinical experience with dronabinol.
http://www.cannabis-med.org/studies…how.php?s_id=90
Dronabinol enhancement of appetite in cancer patients.
http://www.cannabis-med.org/studies…ow.php?s_id=149
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases.
http://www.cannabis-med.org/studies…ow.php?s_id=180
Dronabinol and prochlorperazine in combination
http://www.cannabis-med.org/studies…how.php?s_id=28
Dronabinol enhancement of appetite in cancer patients.
http://www.cannabis-med.org/studies…ow.php?s_id=149
Efficacy of dronabinol alone and in combination
http://www.cannabis-med.org/studies…ow.php?s_id=191
Dronabinol and marijuana in HIV-positive marijuana smokers: caloric intake, mood, and sleep.
http://www.cannabis-med.org/studies…ow.php?s_id=190
Dronabinol and retinal hemodynamics in humans.
http://www.cannabis-med.org/studies…ow.php?s_id=202
Dronabinol reduces signs and symptoms of idiopathic intracranial hypertension
http://www.cannabis-med.org/studies…ow.php?s_id=181
Nausea relieved by tetrahydrocannabinol (dronabinol).
http://www.cannabis-med.org/studies…how.php?s_id=35
Dronabinol in patients with intractable pruritus secondary to cholestatic liver disease.
http://www.cannabis-med.org/studies…ow.php?s_id=116
Treatment of spasticity in spinal cord injury with dronabinol
http://www.cannabis-med.org/studies…ow.php?s_id=112
Cannabinoid Activator Mellows Out Colon
http://www.medpagetoday.com/MeetingCoverage/ACG/tb/4410
Drug testing
Hemp oil causes positive urine tests for THC
http://www.druglibrary.org/crl/drug…0JAnToxicol.pdf
Dystonia
Cannabis sativa and dystonia secondary to Wilson’s disease.
http://www.medscape.com/medline/abstract/15390041
Experiences with THC-treatment in children and adolescents
http://www.cannabis-med.org/studies…how.php?s_id=80
Evaluation of cannabidiol in dystonic movement disorders
http://www.cannabis-med.org/studies…how.php?s_id=14
Cannabidiol in dystonic movement disorders.
http://www.cannabis-med.org/studies…ow.php?s_id=139
Beneficial and adverse effects of cannabidiol in a Parkinson patient
http://www.cannabis-med.org/studies…ow.php?s_id=142
Treatment of Meige’s syndrome with cannabidiol.
http://www.cannabis-med.org/studies…ow.php?s_id=114
———- Page 5
Endocannabinoid Deficiency
Clinical Endocannabinoid Deficiency
http://www.freedomtoexhale.com/clinical.pdf
The endocannabinoid system is dysregulated in multiple sclerosis
http://brain.oxfordjournals.org/cgi…stract/awm160v1
Cannabinoids inhibit neurodegeneration in models of multiple sclerosis
http://brain.oxfordjournals.org/cgi…ull/126/10/2191
Epilepsy
Epilepsy patients are smoking pot
http://www.safeaccessnow.org/article.php?id=1638
CANNABIDIOL TO HEALTHY VOLUNTEERS AND EPILEPTIC PATIENTS
http://web.acsalaska.net/~warmgun/es201.html
Experiences with THC-treatment in children and adolescents
http://www.cannabis-med.org/studies…how.php?s_id=80
Chronic administration of cannabidiol to healthy volunteers and epileptic patients.
http://www.cannabis-med.org/studies…how.php?s_id=42
Anticonvulsant nature of marihuana smoking.
http://www.cannabis-med.org/studies…how.php?s_id=39
Cannabis may help epileptics
http://www.medicalnewstoday.com/articles/4423.php
Hypnotic and Antiepileptic Effects of Cannabidiol
http://www.thecompassionclub.org/me…rue&pageNumber=
Marijuana: an effective antiepileptic treatment in partial epilepsy?
http://www.cannabis-med.org/studies…ow.php?s_id=157
Familial Mediterranean Fever
Pain relief with oral cannabinoids in familial Mediterranean fever.
http://www.cannabis-med.org/studies…how.php?s_id=18
Fertility
Synthetic Cannabinoid May Aid Fertility In Smokers
http://www.medicalnewstoday.com/articles/58063.php
Medical marijuana uses – 700 medical marijuana clinical studies and papers
Fever
A Novel Role of Cannabinoids
http://ccicnewsletter.com/index.php…nfectious_Disea
A Cooling Effect From Cannabis?
http://ccrmg.org/journal/05aut/coolcannabis.html
Fibromyalgia
Delta-9-THC based monotherapy in fibromyalgia patients
http://www.medscape.com/medline/abstract/16834825
Clinical Endocannabinoid Deficiency
http://www.freedomtoexhale.com/clinical.pdf
Cannabis Sativa (Marijuana) for Fibromyalgia
http://www.fibromyalgia-reviews.com/Drg_Marijuana.cfm
THC Reduces Pain in Fibromyalgia Patients
http://www.illinoisnorml.org/content/view/63/35/
Gateway Theory
The Myth of Marijuana’s Gateway Effect
http://www.druglibrary.org/schaffer/library/mjgate.htm
Endogenous cannabinoids are not involved in cocaine reinforcement
http://www.sciencedirect.com/scienc…a4e861a90579fac
No ‘Smoking’ Gun: Research Indicates Teen Marijuana Use Does Not Predict Drug, Alcohol Abuse
http://www.sciencedaily.com/release…61204123422.htm
CLAIM #13:MARIJUANA IS A “GATEWAY” TO THE USE OF OTHER DRUGS
http://www.erowid.org/plants/cannab…is_myth13.shtml
Glaucoma
Marijuana Smoking vs Cannabinoids for Glaucoma Therapy
http://archopht.ama-assn.org/cgi/co…act/116/11/1433
Dronabinol and retinal hemodynamics in humans.
http://www.cannabis-med.org/studies…ow.php?s_id=202
Effect of Sublingual Application of Cannabinoids on Intraocular Pressure
http://www.cannabis-med.org/studies…ow.php?s_id=201
Delta 9-tetrahydrocannabinol in cancer chemotherapy. Ophthalmologic implications.
http://www.cannabis-med.org/studies…how.php?s_id=88
Effect of marihuana on intraocular and blood pressure in glaucoma.
http://www.cannabis-med.org/studies…how.php?s_id=87
Effect of delta-9-tetrahydrocannabinol on intraocular pressure in humans.
http://www.cannabis-med.org/studies…how.php?s_id=40
Marihuana smoking and intraocular pressure.
http://www.cannabis-med.org/studies…how.php?s_id=47
Neuroprotective and Intraocular Pressure-Lowering Effects of (-)Delta-Tetrahydrocannabinol
http://www.unboundmedicine.com/medl…del_of_Glaucoma
Neuroprotective effect of (-)Delta9-tetrahydrocannabinol and cannabidiol
http://www.unboundmedicine.com/medl…f_peroxynitrite
Effects of tetrahydrocannabinol on arterial and intraocular hypertension.
http://www.medscape.com/medline/abstract/468444
Gynocology and obstetrics
Cannabis Treatments in Obstetrics and Gynecology: A Historical Review
http://www.freedomtoexhale.com/russo-ob.pdf
Heart Disease/ Cardiovascular
Marijuana Chemical Fights Hardened Arteries
http://www.webmd.com/heart-disease/…rdened-arteries
The endogenous cardiac cannabinoid system: a new protective mechanism
http://www.cannabinoid.com/boards/thd3x10073.shtml
Cardiovascular pharmacology of cannabinoids.
http://www.biowizard.com/story.php?pmid=16596789
Delta-9-tetrahydrocannabinol protects cardiac cells from hypoxia
http://www.ingentaconnect.com/conte…020001/00002346
Does Cannabis Hold the Key to Treating Cardiometabolic Disease?
http://www.medscape.com/viewarticle/525040_print
Cannabinoid Offers Cardioprotection
http://www.norml.org/index.cfm?Grou…tm_format=print
Heavy Cannabis Use Not Independently Associated With Cardiovascular Risks
http://www.norml.org/index.cfm?Group_ID=6972
Marijuana use, diet, body mass index, and cardiovascular risk factors
http://www.medscape.com/medline/abstract/16893701
Cannabinoids and cardiovascular disease
http://www.unboundmedicine.com/medl…ical_treatments
Cannabinoids as therapeutic agents in cardiovascular disease
http://www.unboundmedicine.com/medl…s_and_illusions
The in vitro and in vivo cardiovascular effects of {Delta}9-tetrahydrocannabinol
http://www.unboundmedicine.com/medl…_oxide_synthase
Cannabinoids prevented the development of heart failure in animal study
http://www.cannabis-med.org/english…el.php?id=145#2
Cannabis use not associated with risk factors for diseases of heart and circulation
http://www.cannabis-med.org/english…el.php?id=225#2
THC protects heart cells in the case of lowered oxygen supply
http://www.cannabis-med.org/english…el.php?id=212#1
Medical marijuana: study shows that THC slows atherosclerosis
http://thenexthurrah.typepad.com/th…al_marijua.html
Cardiovascular Effects of Cannabis
http://www.idmu.co.uk/canncardio.htm
Changes in middle cerebral artery velocity after marijuana
http://www.ncbi.nlm.nih.gov/sites/e…0&dopt=Abstract[/]
—– Page 6
Hepatitis
Moderate Cannabis Use Associated with Improved Treatment Response
http://www.hivandhepatitis.com/hep_…6/091506_a.html
Cannabis use improves retention and virological outcomes in patients treated for hepatitis C
http://www.natap.org/2006/HCV/091506_02.htm
Hepatitis C – The Silent Killer Can Medical Cannabis Help?
http://www.pacifier.com/~alive/cmu/hepatitis_c.htm
Herpes
Cannabis May Help Combat Cancer-causing Herpes Viruses
http://www.sciencedaily.com/release…40923092627.htm
THC inhibits lytic replication of gamma oncogenic herpes viruses in vitro
http://www.pubmedcentral.nih.gov/bo…ml&artid=521080
Suppressive effect of delta-9-tetrahydrocannabinol on herpes simplex virus infectivity in vitro
http://www.ebmonline.org/cgi/content/abstract/196/4/401
Inhibition of cell-associated herpes simplex virus
http://www.ebmonline.org/cgi/content/abstract/185/1/41
The Effect of {Delta}-9-Tetrahydrocannabinol on Herpes Simplex Virus Replication
http://vir.sgmjournals.org/cgi/cont…stract/49/2/427
Hiccups
Marijuana cures hiccups
http://www.yourhealthbase.com/database/a77k.htm
Marijuana For Intractable Hiccups
http://cannabislink.ca/medical/hiccups.html
HIV / AIDS
Marijuana Use Does Not Accelerate HIV Infection
http://paktribune.com/news/print.php?id=139255
THC improves appetite and reverses weight loss in AIDS patients
http://www.cannabis-med.org/studies…ow.php?s_id=189
Dronabinol and marijuana in HIV-positive marijuana smokers: caloric intake, mood, and sleep.
http://www.cannabis-med.org/studies…ow.php?s_id=190
Cannabis in painful HIV-associated sensory neuropathy
http://www.cannabis-med.org/studies…ow.php?s_id=199
Smoked cannabis therapy for HIV-related painful peripheral neuropathy
http://www.cannabis-med.org/studies…ow.php?s_id=172
Short-term effects of cannabinoids in patients with HIV-1 infection
http://www.cannabis-med.org/studies…how.php?s_id=62
Dronabinol as a treatment for anorexia associated with weight loss in patients with AIDS.
http://www.cannabis-med.org/studies…how.php?s_id=21
Effect of dronabinol on nutritional status in HIV infection.
http://www.cannabis-med.org/studies…ow.php?s_id=150
Dronabinol stimulates appetite and causes weight gain in HIV patients.
http://www.cannabis-med.org/studies…how.php?s_id=20
Dronabinol effects on weight in patients with HIV infection.
http://www.cannabis-med.org/studies…how.php?s_id=45
Recent clinical experience with dronabinol.
http://www.cannabis-med.org/studies…how.php?s_id=90
Marijuana as therapy for people living with HIV/AIDS: Social and health aspects
http://www.unboundmedicine.com/medl…_health_aspects
Marijuana and AIDS: A Four-Year Study
http://ccrmg.org/journal/05spr/aids.html
Historical studies
The La Guardia Committee Report
http://www.druglibrary.org/schaffer…lag/lagmenu.htm
Physical, Mental, and Moral Effects of Marijuana: The Indian Hemp Drugs Commission Report
http://www.druglibrary.org/schaffer/Library/effects.htm
MARIAJUANA SMOKING IN PANAMA
http://www.druglibrary.org/schaffer…ama/panama1.htm
The British Pharmaceutical Codex – 1934
http://www.druglibrary.org/schaffer…ical/brit34.htm
ON THE PREPARATIONS OF THE INDIAN HEMP, OR GUNJAH
http://www.druglibrary.org/schaffer…1850/gunjah.htm
DISPENSATORY OF THE UNITED STATES OF AMERICA Fifth Edition (1843)
http://www.druglibrary.org/schaffer…ry/dispensa.htm
New Remedies:Pharmaceutically and Therapeutically Considered Fourth Edition (1843)
http://www.druglibrary.org/schaffer…ry/dunglisn.htm
On the Haschisch or Cannabis Indica
http://www.druglibrary.org/schaffer…ry/bellhash.htm
ON INDICATIONS OF THE HACHISH-VICE IN THE OLD TESTAMENT
http://www.druglibrary.org/schaffer…tory/hashot.htm
The Physiological Activity of Cannabis Sativa
http://www.druglibrary.org/schaffer…istory/japa.htm
CANNABIS, U.S.P. (American Cannabis):
http://www.druglibrary.org/schaffer…ry/vbchmed1.htm
Hormones
Effects of chronic marijuana use on testosterone, luteinizing hormone, follicle stimulating …
http://www.anesth.uiowa.edu/readabs…sp?PMID=1935564
Marijuana: interaction with the estrogen receptor
http://jpet.aspetjournals.org/cgi/c…tract/224/2/404
Huntington’s Disease
EFFECTS OF CANNABIDIOL IN HUNTINGTON’S DISEASE
http://www.druglibrary.org/schaffer…al/hunting1.htm
Nabilone Could Treat Chorea and Irritability in Huntington’s Disease
http://neuro.psychiatryonline.org/c…/18/4/553?rss=1
Hysterectomy
Effect of nabilone on nausea and vomiting after total abdominal hysterectomy
http://www.cannabis-med.org/studies…ow.php?s_id=137
Idiopathic Intracranial Hypertension
Dronabinol reduces signs and symptoms of idiopathic intracranial hypertension
http://www.cannabis-med.org/studies…ow.php?s_id=181
IQ
Findings of a longitudinal study of effects on IQ
http://www.cmaj.ca/cgi/content/full/166/7/887
Heavy cannabis use without long-term effect on global intelligence
http://www.cannabis-med.org/english…el.php?id=115#2
Marijuana does not dent IQ permanently
http://www.newscientist.com/article…ermanently.html
Marinol/Synthetics/ cannabinoid mixtures
CANNABIS AND MARINOL IN THE TREATMENT OF MIGRAINE HEADACHE
http://www.druglibrary.org/schaffer/hemp/migrn2.htm
Marinol vs Natural Cannabis
http://www.norml.org/pdf_files/NORM…al_Cannabis.pdf
The therapeutic rationale for combining tetrahydrocannabinol and cannabidiol.
http://www.medscape.com/medline/abstract/16209908
Unheated Cannabis sativa extracts and its major compound THC-acid
http://www.medscape.com/medline/abs…504929?prt=true
Side effects of pharmaceuticals not elicited by comparable herbal medicines.
http://www.medscape.com/medline/abstract/10394675
Sativex in the treatment of pain caused by rheumatoid arthritis
http://rheumatology.oxfordjournals….bstract/45/1/50
Is dronabinol an effective appetite stimulant?
http://www.cannabis-med.org/studies…ow.php?s_id=188
Sativex in patients suffering from multiple sclerosis associated detrusor overactivity
http://www.cannabis-med.org/studies…ow.php?s_id=168
Sativex® in patients with symptoms of spasticity due to multiple sclerosis
http://www.cannabis-med.org/studies…ow.php?s_id=169
Nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases
http://www.cannabis-med.org/studies…ow.php?s_id=180
Synthetic cannabinomimetic nabilone on patients with chronic pain
http://www.cannabis-med.org/studies…ow.php?s_id=197
Nabilone significantly reduces spasticity-related pain
http://www.cannabis-med.org/studies…ow.php?s_id=200
Sativex produced significant improvements in a subjective measure of spasticity
http://www.cannabis-med.org/studies…ow.php?s_id=170
Analgesic effect of the synthetic cannabinoid CT-3 on chronic neuropathic pain
http://www.cannabis-med.org/studies…how.php?s_id=85
Cannabinoid rotation in a young woman with chronic cystitis
http://www.cannabis-med.org/studies…ow.php?s_id=115
Dronabinol in patients with intractable pruritus
http://www.cannabis-med.org/studies…ow.php?s_id=116
Cannabinoids reduce levodopa-induced dyskinesia in Parkinson’s disease:
http://www.cannabis-med.org/studies…how.php?s_id=54
Nabilone on L-DOPA induced dyskinesia in patients with idiopathic Parkinson’s disease
http://www.cannabis-med.org/studies…ow.php?s_id=153
Nabilone in the treatment of multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=11
Big Pharma’s Strange Holy Grail: Cannabis Without Euphoria?
http://www.counterpunch.org/gardner07082006.html
Sativex showed positive effects in 65 per cent of patients with chronic diseases
http://www.cannabis-med.org/english…el.php?id=230#4
Meige’s Syndrome
Treatment of Meige’s syndrome with cannabidiol.
http://www.cannabis-med.org/studies…ow.php?s_id=114
Migraine/ Headache
CANNABIS AND MARINOL IN THE TREATMENT OF MIGRAINE HEADACHE
http://www.druglibrary.org/schaffer/hemp/migrn2.htm
Dronabinol reduces signs and symptoms of idiopathic intracranial hypertension
http://www.cannabis-med.org/studies…ow.php?s_id=181
Cannabis and Migraine
http://www.pacifier.com/~alive/cmu/…nd_migraine.htm
Clinical Endocannabinoid Deficiency
http://www.freedomtoexhale.com/clinical.pdf
Hemp for Headache
http://www.freedomtoexhale.com/hh.pdf
Chronic Migraine Headache
http://www.druglibrary.org/schaffer/hemp/migrn1.htm
Morning Sickness
Medical marijuana: a surprising solution to severe morning sickness http://www.findarticles.com/p/artic…124/ai_n6015580
Medicinal cannabis use among childbearing women
http://safeaccess.ca/research/cannabis_nausea2006.pdf
Mortality Rates
Marijuana use and mortality.
http://www.pubmedcentral.nih.gov/ar…i?artid=1380837
Marijuana Smoking Doesn’t Lead to Higher Death Rate
http://ccrmg.org/journal/03sum/kaiser.html
How deadly is marijuana?
http://www.medicalnewstoday.com/articles/4426.php
———– Page 7
MS
Sativex in patients with symptoms of spasticity due to multiple sclerosis
http://www.cannabis-med.org/studies…ow.php?s_id=169
Marijuana derivatives may provide MS treatment
http://www.healthypages.net/news.asp?newsid=5381
Marijuana Helps MS Patients Alleviate Pain, Spasms
http://www.mult-sclerosis.org/news/…smsAndPain.html
Cannabis-based medicine in central pain in multiple sclerosis
http://www.neurology.org/cgi/conten…t/65/6/812?etoc
Cannabis-based medicine in spasticity caused by multiple sclerosis
http://www.cannabis-med.org/studies…ow.php?s_id=192
Sativex in patients suffering from multiple sclerosis associated detrusor overactivity
http://www.cannabis-med.org/studies…ow.php?s_id=168
The effect of cannabis on urge incontinence in patients with multiple sclerosis
http://www.cannabis-med.org/studies…ow.php?s_id=185
Nabilone significantly reduces spasticity-related pain
http://www.cannabis-med.org/studies…ow.php?s_id=200
Cannabinoids in multiple sclerosis (CAMS) study
http://www.cannabis-med.org/studies…ow.php?s_id=160
Sativex produced significant improvements in a subjective measure of spasticity
http://www.cannabis-med.org/studies…ow.php?s_id=170
Cannabis-based medicine in central pain in multiple sclerosis.
http://www.cannabis-med.org/studies…ow.php?s_id=175
Do cannabis-based medicinal extracts have general or specific effects
http://www.cannabis-med.org/studies…how.php?s_id=56
Efficacy, safety and tolerability of an oral cannabis extract in the treatment of spasticity
http://www.cannabis-med.org/studies…how.php?s_id=63
cannabis-based extracts for bladder dysfunction in advanced multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=81
Are oral cannabinoids safe and effective in refractory neuropathic pain?
http://www.cannabis-med.org/studies…ow.php?s_id=143
Dronabinol in the treatment of agitation in patients with Alzheimer’s disease with anorexia
http://www.cannabis-med.org/studies…how.php?s_id=61
Cannabis based medicinal extracts (CBME) in central neuropathic pain due to multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=82
Cannabinoids for treatment of spasticity and other symptoms related to multiple sclerosis
http://www.cannabis-med.org/studies…ow.php?s_id=108
Cannabis based medicinal extract on refractory lower urinary tract dysfunction
http://www.cannabis-med.org/studies…ow.php?s_id=103
Analgesic effect of the cannabinoid analogue nabilone
http://www.cannabis-med.org/studies…ow.php?s_id=203
The perceived effects of smoked cannabis on patients with multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=13
Orally and rectally administered delta-9-tetrahydrocannabinol on spasticity
http://www.cannabis-med.org/studies…how.php?s_id=12
Nabilone in the treatment of multiple sclerosis
http://www.cannabis-med.org/studies…how.php?s_id=11
Effect of cannabinoids on spasticity and ataxia in multiple sclerosis.
http://www.cannabis-med.org/studies…show.php?s_id=2
Delta-9-THC in the treatment of spasticity associated with multiple sclerosis.
http://www.cannabis-med.org/studies…show.php?s_id=1
Tetrahydrocannabinol for tremor in multiple sclerosis.
http://www.cannabis-med.org/studies…show.php?s_id=9
Marihuana as a therapeutic agent for muscle spasm or spasticity
http://www.cannabis-med.org/studies…how.php?s_id=53
Cannabis-based medicine in spasticity caused by multiple sclerosis.
http://www.unboundmedicine.com/medl…tiple_sclerosis
Cannabis based treatments for neuropathic and multiple sclerosis-related pain.
http://www.unboundmedicine.com/medl…is_related_pain
The effect of cannabis on urge incontinence in patients with multiple sclerosis
http://www.unboundmedicine.com/medl…ial__CAMS_LUTS_
Can Cannabis Help Multiple Sclerosis? An International Debate Rages
http://www.pacifier.com/~alive/cmu/…bis_help_ms.htm
Cannabis’ Potential Exciting Researchers in Treatment of ALS, Parkinson’s Disease
http://66.218.69.11/search/cache?ei…&icp=1&.intl=us
The endocannabinoid system is dysregulated in multiple sclerosis
http://brain.oxfordjournals.org/cgi…stract/awm160v1
Cannabinoids inhibit neurodegeneration in models of multiple sclerosis
http://brain.oxfordjournals.org/cgi…ull/126/10/2191
Nabilone
The synthetic cannabinoid nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
Nabilone versus prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=120
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Antiemetic efficacy of nabilone and alizapride
http://www.cannabis-med.org/studies…ow.php?s_id=127
Nabilone versus domperidone
http://www.cannabis-med.org/studies…ow.php?s_id=129
Comparison of nabilone and prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=128
Nabilone vs. prochlorperazine for refractory emesis
http://www.cannabis-med.org/studies…ow.php?s_id=146
Nabilone vs. placebo
http://www.cannabis-med.org/studies…ow.php?s_id=156
Effects of nabilone and prochlorperazine on chemotherapy-induced emesis
http://www.cannabis-med.org/studies…ow.php?s_id=131
Superiority of nabilone over prochlorperazine
http://www.cannabis-med.org/studies…ow.php?s_id=126
Nabilone versus prochlorperazine for control of cancer chemotherapy-induced emesis in children
http://www.cannabis-med.org/studies…ow.php?s_id=120
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Effect of nabilone on nausea and vomiting after total abdominal hysterectomy
http://www.cannabis-med.org/studies…ow.php?s_id=137
Nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
Synthetic cannabinomimetic nabilone on patients with chronic pain
http://www.cannabis-med.org/studies…ow.php?s_id=197
Nabilone significantly reduces spasticity-related pain
http://www.cannabis-med.org/studies…ow.php?s_id=200
Nabilone on L-DOPA induced dyskinesia in patients with idiopathic Parkinson’s disease
http://www.cannabis-med.org/studies…ow.php?s_id=153
Nabilone in the treatment of multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=11
Nabilone significantly reduces spasticity-related pain
http://www.cannabis-med.org/studies…ow.php?s_id=200
Analgesic effect of the cannabinoid analogue nabilone
http://www.cannabis-med.org/studies…ow.php?s_id=203
Nabilone in the treatment of multiple sclerosis
http://www.cannabis-med.org/studies…how.php?s_id=11
Comparison of nabilone and metoclopramide in the control of radiation-induced nausea.
http://www.cannabis-med.org/studies…ow.php?s_id=130
Nabilone and metoclopramide in the treatment of nausea and vomiting
http://www.cannabis-med.org/studies…ow.php?s_id=121
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Comparison of the antiemetic efficacy of nabilone and alizapride
http://www.cannabis-med.org/studies…ow.php?s_id=127
Nabilone versus domperidone in the treatment of cytotoxic-induced emesis.
http://www.cannabis-med.org/studies…ow.php?s_id=129
Add-on treatment with the synthetic cannabinomimetic nabilone on patients with chronic pain –
http://www.cannabis-med.org/studies…ow.php?s_id=197
Comparison of bronchial effects of nabilone and terbutaline
http://www.cannabis-med.org/studies…how.php?s_id=43
Nabilone Could Treat Chorea and Irritability in Huntington’s Disease
http://neuro.psychiatryonline.org/c…/18/4/553?rss=1
Nausea
THC improves appetite and reverses weight loss in AIDS patients
http://www.cannabis-med.org/studies…ow.php?s_id=189
Efficacy of dronabinol alone and in combination with ondansetron versus ondansetron alone
http://www.cannabis-med.org/studies…ow.php?s_id=191
Dronabinol and marijuana in HIV-positive marijuana smokers: caloric intake, mood, and sleep.
http://www.cannabis-med.org/studies…ow.php?s_id=190
Nabilone improves pain and symptom management in cancer patients
http://www.cannabis-med.org/studies…ow.php?s_id=177
Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases.
http://www.cannabis-med.org/studies…ow.php?s_id=180
Nausea relieved by tetrahydrocannabinol (dronabinol).
http://www.cannabis-med.org/studies…how.php?s_id=35
An efficient new cannabinoid antiemetic in pediatric oncology.
http://www.cannabis-med.org/studies…show.php?s_id=7
Effect of nabilone on nausea and vomiting after total abdominal hysterectomy.
http://www.cannabis-med.org/studies…ow.php?s_id=137
Marijuana as antiemetic medicine
http://www.cannabis-med.org/studies…ow.php?s_id=134
Efficacy of tetrahydrocannabinol in patients refractory to standard anti-emetic therapy
http://www.cannabis-med.org/studies…how.php?s_id=31
Inhalation marijuana as an antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=155
Nabilone versus prochlorperazine for control of cancer chemotherapy-induced emesis in children
http://www.cannabis-med.org/studies…ow.php?s_id=120
Comparison of nabilone and metoclopramide in the control of radiation-induced nausea.
http://www.cannabis-med.org/studies…ow.php?s_id=130
Nabilone and metoclopramide in the treatment of nausea and vomiting
http://www.cannabis-med.org/studies…ow.php?s_id=121
Nabilone: an alternative antiemetic for cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=123
Comparison of the antiemetic efficacy of nabilone and alizapride
http://www.cannabis-med.org/studies…ow.php?s_id=127
Nabilone versus domperidone in the treatment of cytotoxic-induced emesis.
http://www.cannabis-med.org/studies…ow.php?s_id=129
THC or Compazine for the cancer chemotherapy patient–the UCLA study
http://www.cannabis-med.org/studies…how.php?s_id=34
Comparison of nabilone and prochlorperazine for emesis induced by cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=128
Acute and subacute bronchial effects of oral cannabinoids.
http://www.cannabis-med.org/studies…how.php?s_id=44
Nabilone vs. prochlorperazine for refractory emesis induced by cancer chemotherapy.
http://www.cannabis-med.org/studies…ow.php?s_id=146
Nabilone vs. placebo in chemotherapy-induced nausea and vomiting.
http://www.cannabis-med.org/studies…ow.php?s_id=156
Dose vs response of tetrahydroannabinol (THC) vs prochlorperazine
http://www.cannabis-med.org/studies…how.php?s_id=30 delta 9-
Comparative trial of the antiemetic effects of THC and haloperidol
http://www.cannabis-med.org/studies…how.php?s_id=64
Comparison of delta-9-tetrahydrocannabinol and prochlorperazine.
http://www.cannabis-med.org/studies…show.php?s_id=3
Tetrahydrocannabinol in cancer chemotherapy. Ophthalmologic implications.
http://www.cannabis-med.org/studies…how.php?s_id=88
Antiemetic effect of tetrahydrocannabinol
http://www.cannabis-med.org/studies…show.php?s_id=6
The antiemetic activity of tetrahydrocanabinol versus metoclopramide and thiethylperazine
http://www.cannabis-med.org/studies…how.php?s_id=24
The antiemetic effects of nabilone and prochlorperazine on chemotherapy-induced emesis.
http://www.cannabis-med.org/studies…ow.php?s_id=131
Delta-9-tetrahydrocannabinol as an antiemetic for patients receiving cancer chemotherapy
http://www.cannabis-med.org/studies…show.php?s_id=5
Delta-9-tetrahydrocannabinol as an antiemetic in cancer patients receiving high-dose methotrexate
http://www.cannabis-med.org/studies…how.php?s_id=23
THC as an antiemetic in patients treated with cancer chemotherapy
http://www.cannabis-med.org/studies…how.php?s_id=27
Amelioration of cancer chemotherapy-induced nausea and vomiting by delta-9-THC.
http://www.cannabis-med.org/studies…ow.php?s_id=107
Superiority of nabilone over prochlorperazine as an antiemetic
http://www.cannabis-med.org/studies…ow.php?s_id=126
Antiemetic effect of delta-9-tetrahydrocannabinol in patients receiving cancer chemotherapy.
http://www.cannabis-med.org/studies…show.php?s_id=4
Receptor mechanism and antiemetic activity of structurally-diverse cannabinoids
http://www.unboundmedicine.com/medl…the_least_shrew
Neurons
Marijuana Promotes Neuron Growth
http://www.medpot.net/forums/index.php?showtopic=27460
Marijuana-Like Chemicals in the Brain Calm Neurons
http://www.medpot.net/forums/index.php?showtopic=9686
Marijuana May Spur New Brain Cells
http://www.treatingyourself.com/vbu…read.php?t=5921
Cannabinoids promote embryonic and adult hippocampus neurogenesis
http://www.jci.org/cgi/content/full/115/11/3104
Medical marijuana uses – 700 medical marijuana clinical studies and papers
————— Page 8
Neuropathic pain
Cannabinoids Among Most Promising Approaches to Treating Neuropathic Pain
http://www.redorbit.com/news/health…source=r_health
Cannabis-based medicine in central pain in multiple sclerosis
http://www.neurology.org/cgi/conten…t/65/6/812?etoc
Cannabis in painful HIV-associated sensory neuropathy
http://www.cannabis-med.org/studies…ow.php?s_id=199
Smoked cannabis therapy for HIV-related painful peripheral neuropathy
http://www.cannabis-med.org/studies…ow.php?s_id=172
Two cannabis based medicinal extracts for relief of central neuropathic pain
http://www.cannabis-med.org/studies…ow.php?s_id=143
Cannabis based medicinal extracts (CBME) in central neuropathic pain due to multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=82
Analgesic effect of the synthetic cannabinoid CT-3 on chronic neuropathic pain
http://www.cannabis-med.org/studies…how.php?s_id=85
Smoked cannabis in painful peripheral neuropathy and cancer pain refractory to opiods.
http://www.cannabis-med.org/studies…how.php?s_id=96
Analgesic effect of the cannabinoid analogue nabilone
http://www.cannabis-med.org/studies…ow.php?s_id=203
The perceived effects of smoked cannabis on patients with multiple sclerosis.
http://www.cannabis-med.org/studies…how.php?s_id=13
Cannabis based treatments for neuropathic and multiple sclerosis-related pain.
http://www.unboundmedicine.com/medl…is_related_pain
Neuroprotectant
Marijuana Protects Your Brain
http://www.roninpub.com/art-mjbrain.html
The neuroprotective effect of cannabinoids in a rat model of Parkinson’s disease
http://www.medscape.com/medline/abstract/17196181
Neuroprotective and Intraocular Pressure-Lowering Effects of (-)Delta-THC
http://www.unboundmedicine.com/medl…del_of_Glaucoma
Neuroprotective effect of (-)Delta9-tetrahydrocannabinol and cannabidiol
http://www.unboundmedicine.com/medl…f_peroxynitrite
Neuroprotection induced by Delta(9)-tetrahydrocannabinol in AF5 cells
http://www.unboundmedicine.com/medl…ol_in_AF5_cells
Cannabidiol has a cerebroprotective action
http://www.unboundmedicine.com/medl…iting_mechanism
Cannabidiol but not Delta(9)-THC has a neuroprotective effect without the development of tolerance..
http://www.unboundmedicine.com/medl…nt_of_tolerance
Delta(9)-THC) prevents cerebral infarction
http://www.unboundmedicine.com/medl…ent_hypothermia
Delta(9)-Tetrahydrocannabinol protects hippocampal neurons from excitotoxicity
http://www.unboundmedicine.com/medl…_excitotoxicity
Cannabis and Neuroprotection
http://www.pacifier.com/~alive/cmu/…oprotection.htm
Medical marijuana uses – 700 medical marijuana clinical studies and papers
Nutrition
Oily fish makes ‘babies brainier’
http://news.bbc.co.uk/2/hi/health/4631006.stm
Efficacy of dietary hempseed oil in patients with atopic dermatitis.
http://www.medscape.com/medline/abs…ryText=hempseed
Effects of smoked mar
Alright. That’s enough. Stop spamming my comment section with dozens and dozens of links. I won’t approve any more such “link dumps.”
You mean you won`t have your propaganda countered by overwhelming and irrefutable evidence.
Danman,
Only if you ignore the studies that showed the opposite, such as the following mentioned in the review you cited:
Sorry, last paragraph shouldn’t be blockquoted.
A quick scan through Dim’s copypasta URLs shows a whole lotta advocacy sites and a paucity of actual research articles.
Propaganda, indeed.
@Danman, Orac offers this comment section for *discussion*, not reams of copypasta, especially when you not only haven’t read the articles linked to, you haven’t even read your own copypasta!
Or perhaps you’d like to explain the importance of the following to your “overwhelming and irrefutable evidence” that pot cures cancer:
“Oily fish makes ‘babies brainier'”
Danman, I note that in all the animal studies you’re linking to the animals aren’t being fed cannabis–they’re receiving synthetic cannabinoids by different routes of adminstration (e.g., in one of the Alzhiemer’s cites by direct injection into cerebral ventricles). There’s no evidence in your list supporting your claim “The plant needs to be EATEN …… NOT SMOKED.”
Perhaps you meant to say “The plant needs to be INJECTED DIRECTLY INTO YOUR BRAIN…… NOT SMOKED”?
Danman: THC kills cancer cells by driving ceramide synthesis and apoptosis. That is a fact. I`ve more than made my case.
Krebiozen: Only if you ignore the studies that showed the opposite, such as the following mentioned in the review you cited:
Your confusing two different points, Krebiozen. THC induced Ceramide synthesis and cancer apoptosis remains an unimpeachable fact.
Now as far as Cannabinoid Therapy having paradoxical results, indeed that can be the case. And it is not going work for all patients, all the time, with any cancer. I`m certainly not saying that, nor would the vast majority of even die-hard advocates. I agree with the author that, delivery, dosage, all sorts of details need to be worked out. But always I remind; Cannabis has no known fatal dose. The US gov. has repeatedly tried, and failed to define a lethal dose since forever. Period. That salient fact cannot be overstated. Is there even one conventional cancer regime that has no known lethal dose?
I think one problem actual cannabis oil/medicine patients could, and may in some cases be experiencing is Tumor Lysis Syndrome.
http://en.wikipedia.org/wiki/Tumor_lysis_syndrome
“In medicine (oncology and hematology), tumor lysis syndrome (TLS, alternative spelling tumour lysis syndrome) is a group of metabolic complications that can occur after treatment of cancer,[1] usually lymphomas and leukemias, and sometimes even without treatment. These complications are caused by the breakdown products of dying cancer cells and include hyperkalemia, hyperphosphatemia, hyperuricemia and hyperuricosuria, hypocalcemia, and consequent acute uric acid nephropathy and acute renal failure.”
I also would theorize that the rapid weight loss typically associated with cancer and/or cancer treatment, and breakdown of fatty tissues, and release of toxins within, must exacerbate the problem.
Which brings us back to the THC induced ceramide apoptosis bio-mechanism. A database of patients plotting ceramide blood levels throughout Cannabinoid Therapy will go a long way towards accurate dosing, and tumor status. Especially given often inaccurate conventional tumor markers.
So, Scottynuked ………. I post links to abundant science, much of it peer reviewed, in support of cannabis medicine generally, and all you can come up with is “Dim”?
I would suggest you look in the mirror.
danman, it’s pretty obvious you haven’t even bothered to check out any of those citations in that list. For example, just take a look at the last one on your first spam list, “Marijuana smoking and head and neck cancer.”
Do you think this study is “in support of cannabis medicine”? Think again. In fact, it’s a study examining the association between marijuana use and head and neck cancer RISK, nothing to do with actually TREATING the cancer.
Again, since it’s clear you haven’t actually examined most of these citations, can you point to a single one that demonstrates the CLINICAL efficacy of cannabis use? If not, don’t you agree that more research needs to be done?
You’d have to be crazy to think that no more research is needed into the relationship between cannabis use and mental illness, such as psychosis, for example: http://www.ncbi.nlm.nih.gov/pubmed/24904437 http://www.ncbi.nlm.nih.gov/pubmed/24904437
Orac said; ….. “Uh, Danman, I’m a cancer surgeon AND a cancer researcher.
That explains so much. Your livelihood is in no small way going to be effected. You have a severe conflict of interest here, Dr Orac. How can your position even remotely be taken as unbiased?
For you, and much of the medical/pharma complex, unless the internet is shutdown, there is going be a critical mass of public awareness reached about cannabis medicine/science. You can`t stop it. At that point the DEA/Gov. enforced monopoly the medical/pharma complex on curing is going to fall. And if our wholly corporately owned Gov. decides to try and get tough again, the people will merely return to the black market, or growing it themselves. It`s a genie that cannot be put back in the bottle.
And even if when the research chips fall as they may, and say cannabinoid therapy is shown be roughly as effective as conventional treatments why would one even consider expensive dangerous, toxic chemicals, and radiation bombardment vs. a totally non-toxic plant they can grow in the garden?
Danman,
So how do you explain the studies that found THC-induced cancer cell proliferation and tumor growth? How is that consistent with, “THC induced Ceramide synthesis and cancer apoptosis” or with “confusing two different points”?
As for your claim that:
That isn’t true. The fatal dose is very high compared to a therapeutic (or recreational) dose, but it certainly isn’t unknown. Oral delta-9 THC is actually more toxic (LD50 1,270 mg/kg) than oral acetaminophen (LD50 1,944 mg/kg), which kills hundreds of people every year through accidental overdose.
When you consider hash oil may contain up to 90% THC, a 70kg human might be expected to die after ingesting 5 ounces, which is very unlikely (not to mention unpleasant) but not impossible. IV or inhaled THC is even more toxic, with LD50 of 36–40 mg/kg.
Hyperbolic claims of complete non-toxicity won’t help your cause.
You’re delusional if you think that somehow cannabis will totally upend the standards of care for cancer treatment.
That’s a pretty big “if” there, buddy. So you admit we don’t actually know yet? Don’t you think it’s unethical to tell people that cannabis is a “medical panacea” until we do in fact how effective it is?
AdamG said; ….. “danman, it’s pretty obvious you haven’t even bothered to check out any of those citations in that list. For example, just take a look at the last one on your first spam list, “Marijuana smoking and head and neck cancer.”
Do you think this study is “in support of cannabis medicine”? Think again. In fact, it’s a study examining the association between marijuana use and head and neck cancer RISK” …..
….. “Study: Smoking Marijuana May Prevent Head And Neck Cancers 7/1/14 http://reset.me/story/study-smoking-marijuana-may-prevent-head-neck-cancers/
“Could smoking marijuana really prevent cancer? A recent study suggests that moderate marijuana use is associated with reduced risk of head and neck squamous cell carcinoma (HNSCC), better known as head and neck cancers. Numerous recent studies have shown that cannabinoids have anti tumor effects which most likely explains why marijuana smokers are less likely to get head and neck cancers.
(The Tashkin Study cited previously, shows that even HEAVY CANNABIS SMOKERS had slight decrease in those cancers)
Study conclusion:
Our study suggests that moderate marijuana use is associated with reduced risk of HNSCC. We found that moderate marijuana use was significantly associated with reduced risk of HNSCC. This association was consistent across different measures of marijuana use (marijuana use status, duration, and frequency of use). Further, we observed that marijuana use modified the interaction between alcohol and cigarette smoking, resulting in a decreased HNSCC risk among moderate smokers and light drinkers, and attenuated risk among the heaviest smokers and drinkers.”
Why do you persist in fallacies and mythology that have long ago been dispelled?
I repeat; Since at least 1970 the US Gov. has spent gozillions of dollars trying to demonstrate a cannabis cancer link. If there was, they would be singing it to the heavens on every public service commercial spot they could possibly buy.
It does not exist Adam.
Cupcake, you cited that paper as being “in support of cannabis medicine generally.”
Showing that there is no link between marijuana use and cancer risk says NOTHING about the use of cannabis in cancer treatment, which was the purpose of the list YOU presented.
Danman, now you’re trumpeting a study taht found smoking marijuana for 10 years or more is associated with a reduced risk of HNSCC, when previously you have insisted multiple times that smoking isn’t sufficient and it must be eaten instead to get enough THC exposure to cause significant ceramide synthesis? Clearly you’re just throwing random cites at the wall in the hopes a few might stick.
Danman: “say cannabinoid therapy is shown be roughly as effective as conventional treatments”
AdamG: That’s a pretty big “if” there, buddy. So you admit we don’t actually know yet?
No that was a conservative prognostication. More likely is that Cannabinoid Therapy will be a FAR more effective AND SAFE first approach to most cancers.
And I`m betting these patients will really like the fact their hair won`t fall out, and they won`t spend several hours a day riding the porcelain bus, or have numerous, sometimes debilitating “forever” side effects, even with “successful” conventional treatment, like my in-law. And they`ll just love the fact they won`t have take more DANGEROUS TOXIC FDA APPROVED POISON to counteract the DANGEROUS TOXIC FDA APPROVED POISON to counteract more DANGEROUS TOXIC FDA APPROVED POISON.
And then they`ll be thrilled that instead of LOSING EVERYTHING when they can no longer afford the DANGEROUS TOXIC FDA APPROVED POISON, they can produce an effective, simple medication that at least will do no harm, just by growing a plant in their garden.
The link dump is predictably littered with things that say precisely the opposite of what Danman thinks (as well as trying to play the entiemetic card, which isn’t going anywhere):
The first open-access and self-explanatory. The second isn’t, so let’s turn to a paper that cites it.
You have provided papers that allow the inferences that THC (1) is atherogenic and (2) can promote tumor survival. Well done, Kumar.
JGC said; …… Clearly you’re just throwing random cites at the wall in the hopes a few might stick.
No clearly YOUR confused, or acting stupid.
And, exactly as Orac claimed, you have reached this conclusion based on absolutely no evidence. You simply want it to be true.
Without any clinical trials how can you possibly claim that cannabis treatment of “most cancers” is “FAR more effective”? How will you ever know definitely which cancers it’s less effective for?
Hey nice to smell ya Narad
All hail ….. The Great and Powerful King Turd-Blossom himself.
“Without any clinical trials how can you possibly claim that cannabis treatment of “most cancers” is “FAR more effective”? How will you ever know definitely which cancers it’s less effective for?”
Every single FDA approved poison that killed someone went thru clinical trials. In the long run clinical trials prove nothing, except how much pull a given pharma co has at the FDA.
What a substantial response! I think you’re probably better off sticking to your drug-induced “lucid sleep,” danny boy.
I finally found it!!! The documentary source for the prevailing opinions about cannabis science here at SB,
And it`s all TRUE ….. really …..
AdamG …… “What a substantial response! I think you’re probably better off sticking to your drug-induced “lucid sleep,” danny boy”
LOL!!! ….. you obviously are excluding Turd-Blossom`s cute little patent link in the category of insubstantial responses.
Hmmm, posting lots of links that he had not read and/or comprehended, and random insults. Obviously not a jerk. {/sarcasm}
Actually, because I’m a surgeon, I’d be overjoyed if there were a nontoxic treatment for breast cancer that could replace chemotherapy, which I do not administer. Unless cannabis is truly a miracle cure that could cure cancer without surgery (and you’ve presented exactly zero evidence that it is) surgery will still be required in the treatment of breast cancer. As for my research, there are many other things I could turn my attention to related to cancer. Indeed, I’m already doing that, but mainly because I discovered that putting all my eggs in one basket (i.e., having too narrow a research focus) is a good way to have one’s lab go away when NIH funding gets as tight as it is.
But seriously, a variant of the “pharma shill” gambit? Really? It’s one of the lamest ad hominems there is. I could equally ask: How can your position even be remotely taken as unbiased, given that you so relentlessly have hijacked this thread with dumps of links that don’t prove your point?
This is one that I like to call the fallacy of future vindication. It’s a fantasy antivaccine cranks revel in, and it’s not becoming of you to do the same.
If that were the case, I would embrace cannabis. It’s not, at least not at the moment. There’s a corollary to your statement, though, whether you know it or not, though. It’s best expressed as a question: If the research chips fall where they may and cannabinoid therapy is shown not to have significant anticancer effects, will you abandon your advocacy of it? I’ve already said that I’m willing to go where the science leads, including if it leads to the conclusion that cannabis is the miracle treatment you seem to think it is. Are you willing to go where the science leads if it fails scientific testing?
With a whopping 19 Pubmed entries, 15 with Manuel Guzmán as an author? No, sorry. As I’ve already pointed out, THC itself doesn’t seem to be of much clinical interest in this regard. The receptor system itself? Sure. Weed? Not really.
How do you suppose free-floating ceramide translates into a therapeutic effect? What were the S1P levels?
Adam G beat me to it, but certainly nothing in listss of cites you spammed these comments with suggests “Cannabinoid Therapy will be a FAR more effective AND SAFE first approach to most cancers”.
And I will admit I am confused about one thing–you’ve claimed repeatedly that smoking marijuana isn’t sufficient to acheive a therapeutic exposure level of THC–that it (direct quote) “must be eaten to get enough THC to cause significant ceramide synthesis”.
Yet you’re now claiming that smoking marijuana reduces the risk of developing head and neck squamous cell carcinomas.
So which is it–is smoking it enough, or is eating it an absolute requirement?
Successful completion of clinical trials has nothing to do with “pull”, Danman, and clinical traisl are in fact very good at doing exactly what they’re designed to do: establishing therapeutic window, characterizing safety and demonstrating efficacy.
And they’re great at weeding out failures: for every drug that successfully completes Phase I, II and III clinical trials nine or more other candidates–typically with far greater evidence supporting their potential efficacy than marijuana or cannabinoids have to date–will fail to do so.
There’s certainly no reason to predict that if ever marijuana or marijuana derived cannabinoids ever reach a stage of development where beginning cinical trials is warranted they will turn out to be among the fortunate ten per cent which acheive approval, rather than among the other promising leads that failed to pan out.
How precisely would you consider it to be an “insubstantial” response to your own crowing about the GW patent applications, which promptly blew up in your face because you didn’t understand the difference between an agonist and an antagonist yet again? Remember, I’m the one who actually looked at the list.
It’s not my fault that you’ve been reduced to so much Jell-O with fresh pineapple when your attempts at intimidation didn’t play so well in front of an audience that’s used to posturing blowhards stopping by.
More fun with Danman’s citation list!
“A combined preclinical therapy of cannabinoids and temozolomide against glioma.”
– explores the combined use of TMZ and THC. says nothing about the efficacy of THC or cannabis on its own.
“Simultaneous measurement of three N- acylethanolamides in human bio-matrices using ultra performance liquid chromatography-tandem mass spectrometry.”
– describes a new method to measure the amounts of endocannabinoids in bodily fluids. has absolutely nothing to do with the clinical efficacy of cannabis. A few articles on the list appear to be like this…just measuring endocannabinoid levels.
“Novel hexahydrocannabinol analogs as potential anti-cancer agents inhibit cell proliferation and tumor angiogenesis.”
-I fail to see how results surrounding synthetic hexahydrocannabinol analogs proves anything about the clinical effects of ingested, natural cannabis
“Modulation of the cannabinoid receptors by andrographolide attenuates hepatic apoptosis following bile duct ligation in rats with fibrosis.”
-invovles treating cholestatic liver disease with andrographolide, a natural derivative of a completely different plant with little relation to cannabis. it happens to mention the word ‘cannabinoid’ in the abstract so that’s how it probably got included in your list-of-articles-you-didn’t-read
I could keep going! Just admit it danman, most of your cites have little or nothing to do with your claim that natural cannabis is a “medical panacea.”
I’m aware our host doesn’t like this line of critique, but I’m a say what ya mean type of guy, so:
@Danman
“No clearly YOUR confused, or acting stupid.”
His confused what? He must possess a remarkable thing in this state of confusion, as it’s spelled out extra loud, perhaps for those who can’t read lowercase. Perhaps the acting skill you refer to following is employed to soothe the un-named possession beset with confusion.
Perhaps more cannabis will help you express yourself clearly. Can you copy paste another search result for that?
Don’t bother reading any of the articles from your search. It’s all Big Science tainted stuff, excepting a few sentences which you believe to mean “do moar dope, it’s good for you”.
most of your cites have little or nothing to do with your claim
Ever since our Fearless Box of Blinky Lights banned Gerg the Terminally Clueless it has been a little tame around here.
@Spectator:
I believe – and someone correct me if I’m wrong- but our gracious and magnanimous host only becomes ( righteously, I might add) peeved when his own words are unceremoniously critiqued for minor infractions.
@ shay:
it appears that Gergles has found a new home @ AoA where he serves as a consultant to the brain trust resident at that outlet.
Orac; …. “But seriously, a variant of the “pharma shill” gambit? Really? It’s one of the lamest ad hominems there is. I could equally ask: How can your position even be remotely taken as unbiased, given that you so relentlessly have hijacked this thread with dumps of links that don’t prove your point?”
They do prove my points, and you know it. Your in a state of deep denial.
Doctor, it is simply ludicrous to imply that an industry whose annual cancer treatment worth ALONE is somewhere around 3/4 of trillion annually worldwide would welcome a natural, non-toxic, plant based, and effective treatment with open arms. You have absolutely no integrity whatever if can come back and honestly say there are not intense economic interests involved, and being brought bear on legislators to protect the system.
http://www.cancer.org/aboutus/globalhealth/global-economic-cost-of-cancer-report
“The economic toll from cancer is nearly 20 percent higher than heart disease, the second leading cause of economic loss ($895 billion and $753 billion respectively). This figure does not include direct medical costs, which would further increase the overall economic impact of cancer”
That’s a straw man so friggin’ massive its lighting on fire with your burning stupid can be seen from space. I didn’t say the INDUSTRY would welcome it. I said *I* would welcome it. God, you’re dumb.
Right, you cite a W—pedia article and follow it up with a random, completely unrelated “theory” that amounts to nothing other than the quack-classic “Herxing.”
Hint: TLS has nothing to do with “toxins,” unless you place potassium, phosphorus, and nucleic acids in this category.
For which you provide this:
So, (1) you misread the text and confuse the heart-disease figure to obtain “around 3/4 of trillion [sic]” (2) from a source that explicitly is not talking about medical costs. In order to make a claim about medical industry profit.
Fantastic.
Do you know what these costs represent? Lost years of life and productivity, dumbass. You know, years during which the medical industry could be making money off of people.
@Danman, you need to work on your reading comprehension. More context from the site you yourself cited:
See those words “premature death and disability”? That’s what they’re talking about in terms of economic impact: people who would have been productive tax-paying citizens, but died or became disabled, requiring support from others including governments, due to cancer. That is the cost of foregone productivity. That is *not* the cost of treatment. It even says so! You even quoted it saying so!
He also “trolls” (his words) the Austism Speaks comments using Disqustink.
Danman needs to study up on game theory. Every oncologist may be better off if all oncologists keep quiet about pot* than if they all revealed the secret, but any individual oncologist would be better off if he revealed the secret than if he didn’t.
*And of course all the other mutually exclusive 100% effective, inexpensive, all natural cures for cancer that various interested parties have touted.
“God, you’re dumb” seems like the appropriate reply to all Danman’s drivel.
It’s mildly amusing that he was too out of it not to notice that nearly all of the links were broken in the second dump. Oh, sorry, salvo of “overwhelming and irrefutable evidence.”
^ “too out of it
notto notice”Hint: “TLS has nothing to do with “toxins,” unless you place potassium, phosphorus, and nucleic acids in this category” ….
Narad the Conceptually Dense ….. did you read the word “exacerbate”?
Hint: As in “to exacerbate the problem” (TLS) As a separate, but possibly related issue.
You did see that I used the word toxin relating to fatty tissues, not TLS? Right?
Sweet Jesus ……this is like pulling teeth.
I like his very first line:
Really, if he’s going to copy off a webpage, he could at least look at the *first line*.
And there’s this cite, which you can see clearly focuses on cannabis as a cancer cure:
To this layman, the following doesn’t sound like a *good* thing:
Is salivary secretion something to do with cancer?
I don’t think there’s any cannabis in alcohol:
Those are just the ones I noticed before I gave up and scrolled (and scrolled and scrolled and scrolled) past the rest.
Oh, and he still hasn’t explained the relevance of oily fish and brainier babies to whatever point he’s making.
LW …. “To this layman, the following doesn’t sound like a *good* thing:
Hu, G., Ren, G. & Shi, Y. The putative cannabinoid receptor GPR55 promotes cancer cell proliferation. Oncogene (2010).
Well LW you should have read a bit more …. especially the part about “suggesting a novel cancer biomarker and a potential therapeutic target.”
I guess you missed that.
http://www.ncbi.nlm.nih.gov/pubmed/21057532
“Cannabinoids, the active ingredients in marijuana, have dramatic effects on various organ systems. They exert their effects through two receptor types: CB1, primarily located in the brain, and CB2, primarily located in the immune system. Vertebrates also produce their own cannabinoid-like substances called endocannabinoids, including anandamide and 2-arachidonoylglyceral. Interestingly, some effects of endocannabinoids could not be explained by the signals through either CB1 or CB2. Recently, the orphan G protein-coupled receptor 55 (GPR55) was proposed to be an atypical cannabinoid receptor. In this issue of Oncogene, two groups demonstrated that GPR55 is expressed in various cancer types in an aggressiveness-related manner, suggesting a novel cancer biomarker and a potential therapeutic target.”
Bugger me Danman, you are a prized idiot.
The therapeutic action on this novel target would be to down-regulate GPR55 or make it not respond to cannabinoids in an attempt to reduce its promotion of cancer cell proliferation. Exactly the opposite of what you are suggesting.
“I guess you missed that.”
That would be because there was no link and it’s difficult for me to search on an iPhone with a barely functional Internet connection. Maybe it was in that list of links that showed up after scrolling thirty or so iPhone pages.
I did read the article about oily fish and brainier babies, did you?
LW:
I have a friend who stays far away from marijuana. She grew up in the 1960s/70s with the “cool” parents that regularly toked with friends. She said they would have what they thought were “brilliant” conversations, and all she heard was nonsensical drivel.
She made the decision before entering high school that she preferred to keep her brain cells intact.
Speaking of exacerbation, this was an excellent addition to the Gish gallop:
Cabral, G. A. & Marciano-Cabral, F. Cannabinoid-mediated exacerbation of brain infection by opportunistic amebae. J Neuroimmunol 147, 127-130 (2004).
Because sludging up microglia and inhibiting TNF-α is a really good idea for treating brain cancers.
Anyway,
OK, you have pulled TLS out of your ass and concluded that a “release of toxins” that you have also pulled out of your ass “must exacerbate the problem.” How? What “toxins”? What’s “exacerbated”? What happened to the “simple medication that at least will do no harm”?
That’s because he still thinks anything that has anything to do with cannabinoid receptors means “Weed!” He’s gotten this backward repeatedly.
How do you inhibit sphingosine kinase-1 and glucosylceramide synthase?
Apparently if you smoke enough weed, you see the sentence “suggests a potential therapeutic target” in a journal article but your mind interprets it as “CANCER CURE!”
That’s some powerful stuff.
Danman @186
You mean like taxol? Oh, the industry did welcome it. Natural, plant-based, and effective. I hope you can allow that a treatment which needs to kill cancer cells in order to be effective cannot be “non-toxic” ̶ it has to be toxic to the cancer cells.
Danman, considering that you cannot seem to understand why your arguments are not working on this crowd I’d ask what you’ve been smoking, but I guess given the topic that’s obvious.
ChrisP: …… “Bugger me Danman, you are a prized idiot.”
“The therapeutic action on this novel target would be to down-regulate GPR55 or make it not respond to cannabinoids in an attempt to reduce its promotion of cancer cell proliferation. Exactly the opposite of what you are suggesting.”
Get a carrot and bugger yourself ….. The point I have all along stressed is that CB1 and CB2 are the receptors that are known to be involved with THC induced ceramide synthesis and apoptosis.
Nowhere did I imply that in order to treat cancer GPR55 should be up or down regulated.
And ……. “Whether the orphan G-protein coupled receptor GPR55 is also a cannabinoid receptor remains unclear as a result of conflicting pharmacological studies” ….
http://www.ncbi.nlm.nih.gov/pubmed/19723626
“Nowhere did I imply that in order to treat cancer GPR55 should be up or down regulated.”
Then why was the article in your gish gallop — oh, I mean overwhelming and irrefutable evidence list?
God, you’re dumb.
Let’s review:
To this layman, the following doesn’t sound like a *good* thing
Well LW you should have read a bit more …. especially the part about “suggesting a novel cancer biomarker and a potential therapeutic target.”
I guess you missed that.
Really, were you saying that it’s a good thing because you thought eliminating activation of the cannabinoid receptors fits right in with your hypothesis?
^ F*cking blockquotes.
Chemmomo; …. “welcome a natural, non-toxic, plant based, and effective treatment …….You mean like taxol?
No I meant cannabis, specifically THC. Cannabis has no known lethal dose. I`m guessing Taxol does.
You also apparently didn`t read, or don`t believe Dennis Hill?
http://www.cureyourowncancer.org/how-cannabis-oil-works.html
“First let’s look at what keeps cancer cells alive, then we will come back and examine how the cannabinoids CBD (cannabidiol) and THC (tetrahydrocannabinol) unravels cancer’s aliveness.
“In every cell there is a family of interconvertible sphingolipids that specifically manage the life and death of that cell. This profile of factors is called the “Sphingolipid Rheostat.” If endogenous ceramide(a signaling metabolite of sphingosine-1-phosphate) is high, then cell death (apoptosis) is imminent. If ceramide is low, the cell is strong in its vitality.
Very simply, when THC connects to the CB1 or CB2 cannabinoid receptor site on the cancer cell, it causes an increase in ceramide synthesis which drives cell death. A normal healthy cell does not produce ceramide in the presence of THC, thus is not affected by the cannabinoid.
The cancer cell dies, not because of cytotoxic chemicals, but because of a tiny little shift in the mitochondria. Within most cells there is a cell nucleus, numerous mitochondria (hundreds to thousands), and various other organelles in the cytoplasm. The purpose of the mitochondria is to produce energy (ATP) for cell use. As ceramide starts to accumulate, turning up the Sphingolipid Rheostat, it increases the mitochondrial membrane pore permeability to cytochrome c, a critical protein in energy synthesis. Cytochrome c is pushed out of the mitochondria, killing the source of energy for the cell.”
Funny, you’ve already been provided an LD50. THC is more toxic in rats than taxol (PDF).
Now you’re simply ignoring the fact that you’ve been provided with multiple examples of cancer cells that do exactly the opposite and trying to start over.
Narad; ……. Funny, you’ve already been provided an LD50. THC is more toxic in rats than taxol (PDF).
Oops ….. Page Not Found!
Hmm, we couldn’t find the page you’re looking for. Care to try a search?
If you had used your remaining neurons, you could have easily enough found the payload in the link.
Then again, given that you’ve recently posted scores of broken links, I can see why that might have been a bit much for you.
I hate to break this to you, but this is the effect of a cytotoxic chemical. Moreover, you’re stuck with the Warburg effect.
Danman quoting Dennis Hill: ….. “Very simply, when THC connects to the CB1 or CB2 cannabinoid receptor site on the cancer cell, it causes an increase in ceramide synthesis which drives cell death.”
Narad: …. Now you’re simply ignoring the fact that you’ve been provided with multiple examples of cancer cells that do exactly the opposite and trying to start over.
Wait a minute …….. so what your saying is Dennis Hill is a liar? Didn`t have cancer? Didn`t cure it with cannabis oil? Has no idea what he`s talking about?
Go ahead, lets hear it.
Narad ….. “The cancer cell dies, not because of cytotoxic chemicals, but because of a tiny little shift in the mitochondria.”
I hate to break this to you, but this is the effect of a cytotoxic chemical.
….. so your saying the man did chemo? The man is a liar?
That exactly what I thought. Too f*cking cowardly to backup your own convictions.
I`m done here. I`ve made my points.
Danman, obviously the expression I should have used was Bugger me sideways.
You are a doubly prized idiot.
A hint, an excavator is a better tool for digging large holes than a teaspoon.
Do keep providing the chuckles.
You mean that Dennis Hill? Good thing he has an MBA, I bet he’s great at making money off fools like you. Speaking of shills, did you notice that the first thing you see on his website is an ad for his own tshirts?
He even has his very own page on whale.to! That’s how you know he’s legit.
Oh, it gets better. In the most popular video of Mr. Hill (https://www.youtube.com/watch?v=WRJxauzFfNM) he starts off by saying
You think he would’ve mentioned the ten years at MD Anderson!
Of course, he’s not near brave enough to mention an association with either Baylor or MD Anderson on his LinkedIn page.
I’m talking about you, jackass. You were trying to reboot the screaming failure of link dumps by cycling back to where you started.
Maybe he’s an imbecile. Nobody’s shown whether the intrinsic or extrinsic pathway is involved even if the ceramide routine were to hold water, but do you “think” that using a chemical to get mitochondria to disgorge cytochrome c to kill their cells is some magic thing that only the Goddess Herb can do? Oops.
Oh, dear, Bitsy does have some impulse control issues, doesn’t he?
Do stick the flounce. And see if you can trade a bag for some apostrophes.
Narad:
No no no no, it’s not cytotoxic! It just kills the cell. Totally a different thing, right?
First of all, to those who posted about prohibition: the belief that prohibition was a total failure is oversimplified and as a group of critical thinkers, you can do better. Look up the research. Just because it’s part of our national mythology doesn’t make it true.
Secondly, and more importantly, thank you for the interesting discussion. My dh has stage iv lung cancer (adeno). He had a complete response to first line doublet chemo and is currently NED. I’m sure glad he did the govt. approved “poison”. We are one of the lucky 1-3% with a complete response, but I know many, many more who’ve extended their lives and eased their symptoms with chemo.
I look forward to reading more and await your article on cannabis oil and cancer. I’m looking for ideas when chemo options run out as they tend to do with stage iv.
PollylovesJoe: First of all, to those who posted about prohibition: the belief that prohibition was a total failure is oversimplified and as a group of critical thinkers, you can do better. Look up the research. Just because it’s part of our national mythology doesn’t make it true.
During Prohibition, consumption of alcoholic beverages actually rose, the number of deaths related to alcohol poisoning rose, organized crime expanded and became more deadly than ever, and a number of cities openly flouted federal law. To me, that smacks of failure. If Prohibition had any actual success, I’m unaware of it. I think you might be using a different dictionary or history than the rest of us use.
PGP, you’ve unsurprisingly fallen for the “national mythology” just as PollylovesJoe pointed out. A simple read of the wiki on US Prohibition would have told you that your claim above is not based in reality, and would’ve lead you to this source: http://www.nber.org/papers/w3675.pdf
It pays to search before you type, PGP.
As Hilll hasn’t offered any evidence that the cannabis oil protocol he followed caused his cancer to remit bu seems to simply be embracing a post hoc ergo procter hoc logical fallacy (seasoned liberally with “What else could it be?”), and given the five year survival rates of untreated patients presenting with the same stage of prostate cancer, no: I don’t believe his claim that his cancer was cured by cannabis oil–he’s given me no reason to.
AdamG: Note that the paper itself says that the stats they’re working off of are of dubious reliability. Even if consumption fell, my other three points are true.
Cirrhosis of the liver rates went down which does tend to indicate an overall decline as it tends to be dose dependent..
In any case I think the social issues with crime were much more what any failure was about even if the number of people who drank went down or the number of drinks per person per year went down.
And both those numbers, IMO could go down substantially and alcohol poisoning go up. Just the standard issue human foolishness of the I can’t get it often so when I get it I better binge on it variety added to changes in distillation (or just greater variation in home distillation rather than large scale industrial) could have you end up with more poisoned people (assuming if those stats are purely ethanol and not including people drinking impure spirits) even with less overall drinking.
Knowing you I doubt you’ve actually bothered to research these points before stating this.
The claim is for death rates, not prevalence. Take a look at Figures 2 and 3 in Dills & Miron; apparently, Prohibition magically immediately fixed the number of cirrhosis deaths but didn’t cause any further decrease. The hypothesis of simple dose dependence is also belied by the the other-country data.
I’m not going to go through and look at how much they massaged the data, but I’d be a lot more confident in this conclusion if it were coming from epidemiologists rather than economists.
Kaymarie: There was a lot of alcohol poisoning during Prohibition, usually from impure spirits or people trying to drink alcohol not intended for human consumption (like wood or rubbing alcohol). Maybe the rates of cirrhosis went down because heavy drinkers either tried to extend their stock by drinking less, or they succumbed to other causes.
AdamG: Did you ever read Last Call? Or other actual historical accounts of Prohibition? I’d rather rely on historians than economists, who are to a man ignorant of history and tend not to get out in the fresh air.
[…] stated my position on marijuana last time, which is that marijuana should be at least decriminalized or, preferably, legalized, taxed, and […]
You are speaking very well from a scientific,western medicine model. Marijuana is an herb and so expands out from that model and can not be understood through the limited double blind placebo test process.
Traditional herbalism has always included relationship in its equations, as relationship plays a big part in health and wellness. The relationship between practitioner, patient and tool used is multidimensional and can thus far is not well addressed by western model research, so those that think in that model, can not truly appreciated the multidimensional benefits of not just “weed” as you stated, but all weeds.
The idea that just because you can standardize a chemical to induce a response on a percentage of patients and may cause a multitude of side effects, makes it better to take than an herb that has constituents from nature and recognized by the body and used for centuries, so the body knows what to do with it and is in relationship, for me doesn’t work. I have studied in traditional herbalism many years and hold a masters in herbal studies (a western perspective model). I love the science of phyto chemicals and all the left brained stuff. But it is only one side of the equation. Herbalism opens to the left brain too, relationship, change, growth, evolution, where most of us live and at least have experience. We don’t have double blind placebo tests to help us navigate. But we do have herbs, the plants that have accompanied us for ever. Thank goodness.